 Welcome to HRA Latest with news and updates for those working or involved in health and social care research.

This week was a landmark moment for the health and social care research sector with the updated clinical trials regulations coming into force on Tuesday. There are some important changes, so to make sure you're ready we've shared a round-up of our work on the regulations, operational updates and everything you need to know.
With the changes to the regulations in mind, I've also written about the important role of our Research Ethics Committees and the people who serve on them. They continue to play a vital role in ensuring people have the opportunity to take part in research they can trust.
This month, we have also published two big pieces of data. The first is our UK-wide performance data for 2025, which looks at things like the number of applications we reviewed and how long those reviews took. The second is our latest research transparency data. We've reported on how UK clinical trials in 2023 and 2024 performed against our policy expectations, which are now a legal requirement of the new clinical trials regulations.
We're also sharing our response to the reduction in average UK clinical trial set-up time to 122 days, which represents significant progress against the UK government's 150 days target.
For regular updates and the latest news, you can also follow us on LinkedIn.
Matt Westmore, Chief Executive
|
  New legal requirements for clinical trials came into force on 28 April 2026
Working in partnership with the Medicines and Healthcare products Regulatory Agency we have sought the views of patients, researchers, healthcare professionals and industry to inform and develop the new regulatory reforms.
The updated regulations are designed to protect trial participants, strengthen patient safety, and accelerate approvals by reducing unnecessary burdens on researchers, to support high-quality, trusted research taking place in the UK.
Visit our website for a round-up of our work on the regulations and the operational and guidance updates we've made to support the research community.
 Applications reviewed faster than ever in 2025
The review and approval of applications for health and social care research in the UK is happening faster than ever according to a review of our data from 2025.
We've taken a look back at our performance data from 2025 which showed that the total number of applications reviewed by Research Ethics Committees in the UK increased by 3.1% last year.
The time taken for the reviews for these applications to be completed reduced to a record low of 29 days for commercially sponsored studies in 2025 – less than half of our target of 60 days.
 Blog: making it clearer how RECs review research
The updated clinical trials regulations strengthen how studies are reviewed and approved, including clearer expectations for the constitution of Research Ethics Committees (RECs).
Our Chief Executive, Matt Westmore, has written a blog about how RECs review research, the ordinary people who sit on them and how their decisions are grounded in expertise and real-world experiences.
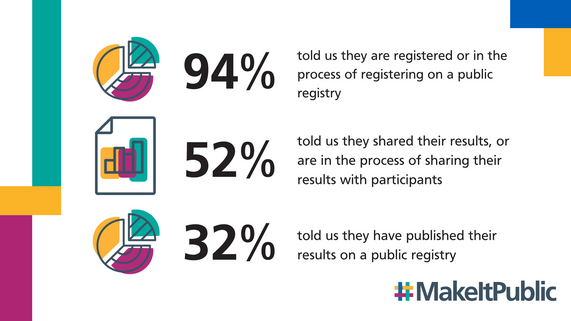 Latest research transparency data shows record number of studies registered
This month, we published our latest research transparency data. It covers the registration status of clinical trials in the UK from 2024, and whether trials that closed in 2023 have published a summary of results on a public registry within 12 months of the trial ending and shared a summary of results with their participants.
It shows that more studies than ever are registered on a public registry, and that more than half of studies completed in 2023 told us they shared their results with participants. However, two thirds of studies haven't published, or told us they've published, their results on a public registry.
We're sharing this data all together for the first time to give a full picture of compliance with policy requirements and expectations for research transparency, which is now a legal requirement under the new clinical trials regulations for clinical trials of medicines.
 Important milestone for UK clinical trials
The average set-up time for commercial clinical trials in the UK has fallen to 122 days, down from 169 during the same period last year, according to new data from the Department of Health and Social Care.
The reduction in average set-up time marks significant progress against the Prime Minister's target of 150 days, which was set out in the Life Sciences Sector Plan last year.
We are proud to have played a key role in helping meet this target, which has only been made possible by the entire health and social care research system in the UK coming together.
 UK Biobank data breach
Last week it was reported that data from UK Biobank had been advertised for sale online.
The UK Biobank is the world’s most comprehensive biomedical dataset, holding the records of more than half a million volunteers. It was given Research Ethics Committee (REC) approval in April 2021.
Access to UK Biobank for researchers is granted by the biobank, based on the wider REC or CAG approval the biobank had already received, and subject to a rigorous vetting process. Following this process is a condition of REC approval.
We are currently investigating whether a serious breach has occurred based on the REC approval for the study.
 Guidance to support organisations that identify research participants
We've published new guidance to support sponsors, NHS and Health and Social Care (HSC) organisations to set-up and manage Participant Information Centres (PICs).
PICs are NHS and HSC organisations, such as NHS Trusts, which identify participants for research studies but do not undertake the research.
The new guidance is part of our ongoing work to improve study set-up.
 NIHR public partnerships action plan launched
The National Institute for Health and Care Research (NIHR) has published a new action plan for delivering its strategic commitments for public partnerships.
As part of the plan, we will work with NIHR and other key partners to discuss and identify strategic priority areas where cross-system leadership and action would strengthen public involvement across the health and social care research sector.
The plan also refers to working with the Charities Research Involvement Group and HRA on developing a core common patient and public involvement question‑set for application forms. This aims to make it easier for researchers to provide the information that reviewers of funding committees, Research Ethics Committees and the Confidentiality Advisory Group need.
 Building better research services
This week, we have released new functionality onto our digital service, plan and manage health and care research, that means we can now accept modifications for eligible studies taking place across more than one nation for the first time. We have also introduced improvements to the user experience as part of this release.
Plan and manage health and care research will digitise the end-to-end research journey, allowing users to plan and prepare new research, make changes to existing research and review and approve research applications in a seamless way.
At this early stage, we are working closely with a small group of sponsors who are using the new service. To keep up-to-date with our progress and our plans to gradually open up the service to more users, please sign up for the .
 News from across UK research
Below are some updates you may have missed about health and social care research in the UK:
|