|
Having trouble viewing this email? View it as a Web page.
SEPT. 21, 2021
|
Hannah is back to answer some of the most frequent questions we're hearing about COVID-19 and testing:
- I'm fully vaccinated -- do I still need to get tested?
- When should I be tested?
- What if I test positive?
Find a COVID-19 testing site near you.
|
|
 |
The Virginia Department of Health announced the addition of QR codes – a type of barcode that can be scanned with smartphones – to Virginia COVID-19 vaccination records.
These allow anyone to show proof of vaccination with a digital or printed QR code instead of a paper card, and without the need for an app. QR codes will help improve the consistency and security of vaccination information while protecting individual privacy.
A person vaccinated in Virginia can visit vaccinate.virginia.gov to obtain their free vaccination record with QR code, which can then be saved to a phone gallery, printed on paper, or stored in a compatible account.
The U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) have not yet issued guidance on whether the general public should get COVID-19 vaccine booster doses.
Statement from Virginia State Vaccination Coordinator Dr. Danny Avula: "An advisory committee of the U.S. Food and Drug Administration (FDA) voted in favor of recommending booster doses of the Pfizer-BioNTech COVID-19 vaccine to Virginians 65 years and up and those at high risk for severe COVID-19. This is simply a recommendation at this point, and booster doses will not be available in Virginia until the FDA issues an updated authorization, and the Centers for Disease Control and Prevention (CDC) issues new guidance."
Arlington County will continue to coordinate with VDH and will provide more information about booster doses when more guidance is available.
The Pfizer vaccine is currently available and FDA authorized for those ages 12 years and up.
Having the recently release Pfizer data available for younger children (ages 5-11) is the first step. To become available to the public for use, it will still need to be authorized by the FDA and then recommended for use by CDC’s Advisory Committee on Immunization Practices (ACIP).
If FDA deems this is an emergency situation for children, they could issue an EUA a few weeks after the application has been submitted -- for the Pfizer vaccine, the timing of this could be as early as the end of October, but there are no dates available at this time.
In the first week of June 2021, Moderna submitted data for expansion of their vaccine EUA, to include 12 to 17 year olds. FDA and ACIP review of these new data still need to occur.
Please see the recent statement titled “FDA Will Follow The Science On COVID-19 Vaccines For Young Children”.
Questions about COVID-19? Call the County's public information line at
703-228-7999, Mon.-Fri., 9 a.m.-5 p.m.
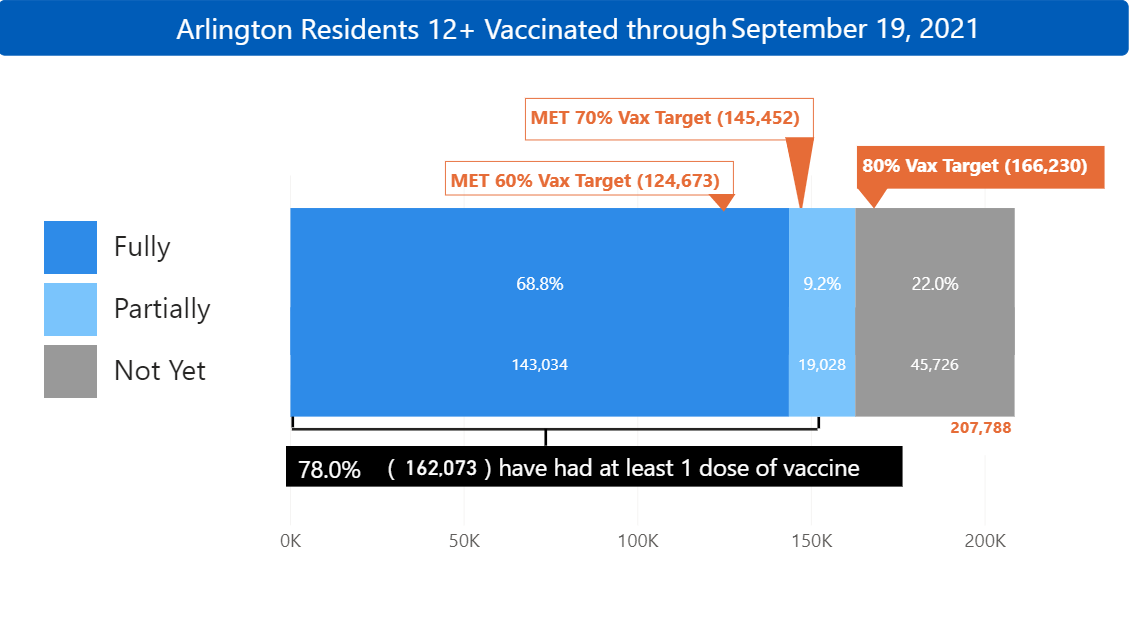
78% of Arlington residents 12 years and older have received at least one dose of a COVID-19 vaccine, and more than 68% are fully vaccinated.
Visit the Arlington COVID-19 Vaccination Dashboard to view more detail on vaccination progress, including distribution by race/ethnicity of residents 12+ vaccinated with at least 1 dose.
|