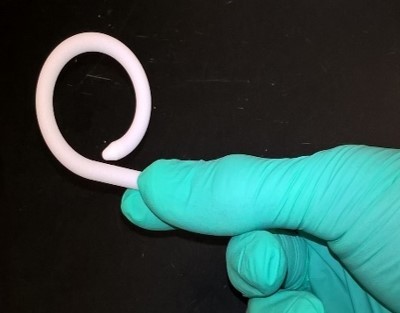
First-in-human clinical trials are underway for the UroMonitor, a wireless catheter-free intravesical pressure sensor that enables bladder monitoring - a FitBit for the bladder. Margot Damaser, PhD and Steve Majerus, PhD jointly create and oversee development of novel wireless catheter-free monitoring devices within the APT Center.
Urodynamics (UDS) is the gold-standard method of quantitatively evaluating the ability of the lower urinary tract to efficiently store and empty urine. However, performing UDS poses numerous challenges including requiring specialized equipment and staff as well as causing patient discomfort. The flexible UroMonitor is shaped like a coil and inserted into a patient’s bladder lumen. It tracks physiological data over a 4-7 day monitoring period and wirelessly transmits the information to the patient’s electronic medical record to inform diagnosis and clinical management. The patient can then remove the device by a string and dispose of it completely.
The primary objective of the human subject study is to assess comfort, and ease of insertion and extraction of the UroMonitor in women with overactive bladder. The secondary objective is to assess UroMonitor functioning and ability to measure pressure in the bladder and wirelessly transmit it reliably to a receiving radio outside the body.
Results of the first three subjects show the UroMonitor:
- Can be inserted and extracted safely without difficulty and with minimal pain in women
- May initially exacerbate OAB symptoms but subjectively causes minimal patient discomfort during testing and after removal
- Data showed a strong correlation to measured catheter pressures and voiding behaviors
To the team's knowledge, this is the first example of wireless catheter-free bladder pressure data collection in humans. The UroMonitor enables ambulatory bladder pressure data collection without catheters or wires impeding normal activity. Preliminary results indicate the UroMonitor is able to identify urodynamically relevant bladder events. The device is easily inserted and removed in ambulatory female patients and does not cause voiding obstruction or any subjective discomfort while in place.
The team is led by Dr. Damaser, Senior Research Career Scientist at Louis Stokes Cleveland VA Medical Center, Full Staff in the Department of Biomedical Engineering in Lerner Research Institute at Cleveland Clinic, and Professor of Molecular Medicine at the Cleveland Clinic Lerner College of Medicine and Biomedical Engineering at Case Western Reserve University, and includes Steve Majerus, PhD, APT Center Investigator and Research Scientist at Cleveland VA, and Samir Derisavifard, MD and Howard Goldman, MD, from Cleveland Clinic Glickman Urological and Kidney Institute.
|