Cancer Types of Patients in the Cancer Moonshot Biobank
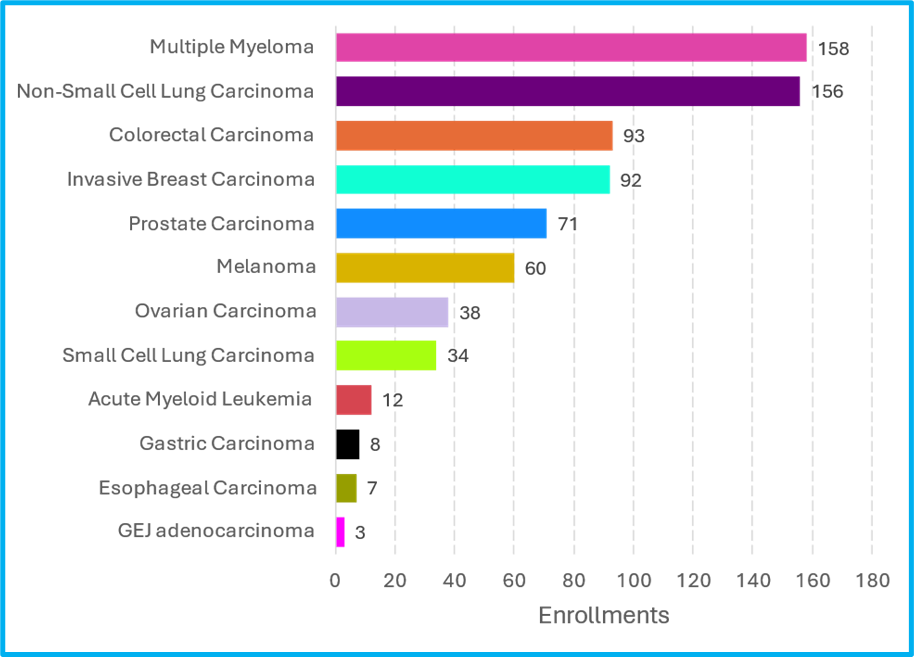 To date, 732 participants have enrolled into the Cancer Moonshot Biobank. Multiple Myeloma and Non-Small Cell Lung Carcinoma are the most common cancer types of our enrolled participants. Participants with Colorectal Carcinoma and Invasive Breast Cancer are also well represented in the study.
Engagement Corner
Virtual Office Hours – Held on Two Thursdays Per Month
Cancer Moonshot Biobank study staff hold virtual office hours two Thursdays a month from 12 p.m. to 12:30 p.m. Eastern Time. Drop in at any time over the half hour to ask questions about the protocol, Rave forms, and any other questions. The office hours are not intended to replace phone calls and emails for sites’ urgent questions, but to provide a regular time when sites know study staff are available. Reminders are sent before the office hours.
Click the TEAMS MEETING LINK to join.
Individual 1:1 Meetings Are Available to Answer Your Protocol Questions
Cancer Moonshot Biobank study staff are happy to schedule an individual meeting with your site staff to answer any questions about the protocol. Please contact Dr. Jane Wanyiri (jane.wanyiri@nih.gov) to schedule a meeting.
|
Celebrating Our Outstanding Sites
Congratulations to four clinical sites for recently enrolling their first patient into the Cancer Moonshot Biobank!
- Aurora Saint Luke’s Medical Center
- Edwards Comprehensive Cancer Center
- Hartford Hospital
- LincolnHealth – Miles Campus and Hospital
What’s New in CTSU:
Monthly Meeting Slides
January 2025 Meeting Slides
|
Questions and Answers
Question 1: If a patient with multiple myeloma had a bone marrow transplant, does that make them ineligible for the study?
Answer 1: No, a bone marrow transplant doesn’t make the patient ineligible. Most patients with multiple myeloma have had a bone marrow transplant. The most important thing is to enter the information about the bone marrow transplant in Rave.
******************************************************************************
Question 2: An enrolled patient on an Appendix A medication has now progressed, and their provider is changing their treatment to chemotherapy based on the patient’s scans. The patient didn’t have a biopsy and doesn’t want to undergo another biopsy at this time. If we can’t acquire a progression biopsy, does the patient need to be removed from the study?
Answer 2: The study team was consulted for this patient, and they recommended keeping the patient on-study, collecting progression blood before starting the chemotherapy, and following the patient to continue to collect blood samples and clinical data. Communicating the value of progression biopsy and blood samples to the participant is vital, but we understand that progression may be a sensitive time for patients and biopsies may not always be an option.
******************************************************************************
Question 3: If a patient is receiving a biosimilar of an Appendix A therapy, are they still eligible for enrollment? For example, we have a patient on FOLFOX + bevacizumab-awwb; does the fact that they are on a biosimilar exclude them?
Answer 3: Biosimilars are allowed. The data entry would be under the original drug. In this case, it would be bevacizumab.
******************************************************************************
Question 4: Is a patient with stage IV rectal cancer eligible? What about a patient with cecum cancer?
Answer 4: Yes, both patients would be eligible under the colorectal cancer designation of the study.
Reminders
- The Cancer Moonshot Biobank study will cover the cost of consent form translations and IRB approval. All site-specific main consent forms are translated by default into Spanish and loaded into eConsent application.
- Thank you for providing updated contact information for lead CRAs, new CRAs, and site addresses. Updated information is vital for study staff to provide timely access to eConsent, the Engagement website, newsletters, and monthly meetings.
- For those sites that have not responded to Dr. Jane Wanyiri’s request, please send the updated contact forms to jane.wanyiri@nih.gov.
- Update the roster through Regulatory Support Services (RSS).
|