| |
|
If your email program has trouble displaying this email or images, view as a webpage.
|
|
|
Back to TOP

White House Launches New Indo-Pacific Economic Framework
The Biden administration recently launched the Indo-Pacific Economic Framework for Prosperity (IPEF) with 13 trading partners: Australia, Brunei, Fiji, India, Indonesia, Japan, Republic of Korea, Malaysia, New Zealand, the Philippines, Singapore, Thailand, and Vietnam.
The framework is intended to achieve high-standard commitments with these countries that will deepen U.S. economic engagement in the region. Trade with the Indo-Pacific region supports more than 3 million U.S. jobs and is the source of nearly $900 billion in foreign direct investment in the United States. With 60% of the world’s population, the region is projected to be the largest contributor to global growth over the next three decades.

“The United States and our partners in the region believe that much of our success in the coming decades will depend on how well governments harness innovation — especially the transformations afoot in the clean energy, digital, and technology sectors — while fortifying our economies against a range of threats, from fragile supply chains to corruption to tax havens,” a White House fact sheet said. “The past models of economic engagement did not address these challenges, leaving our workers, businesses, and consumers vulnerable.”
The framework focuses on four key pillars: supply chain resilience, clean energy, fair economic policies, and trade — including commitments to good regulatory practices (GRP) within the digital economy.
The Office of the U.S. Trade Representative leads the trade pillar while the U.S. Department of Commerce helms the other three. The FDA’s Office of Trade, Mutual Recognition, and International Arrangements is involved with the trade pillar because it concentrates on GRPs, trade facilitation, and sanitary and phytosanitary measures, which are rules and procedures used by governments to ensure that foods and beverages are safe to consume, and that animals and plants are protected from pests and diseases.
Designed to be flexible, the framework gives participating countries with varying levels of regulatory maturity opportunities to collaborate. IPEF partners may choose not to participate in all four pillars, but once a partner commits to pillar discussions, it cannot opt out of individual commitments within a pillar.
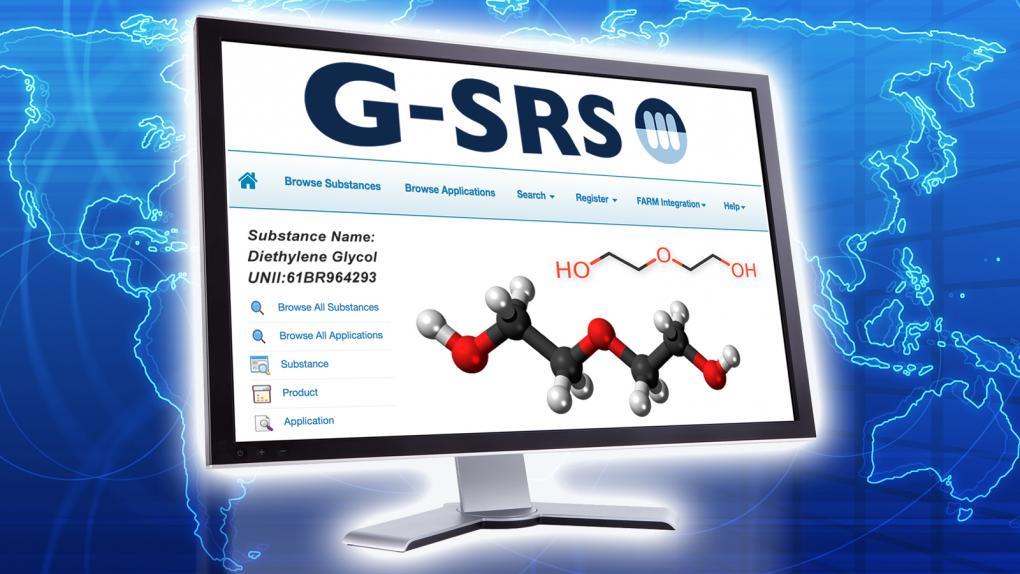
FDA, NIH, and EMA Launch Global Substance Registration System
As the world’s economies become increasingly interdependent, globalization is fueling a steady rise in the number of imported FDA-regulated food, medical, tobacco, and cosmetic products. At the same time, an increasing number of clinical trials are being conducted abroad to save money and ensure a more diverse patient population. To address the public health impacts of these global shifts, the FDA has been working closely with the National Institutes of Health (NIH) and the European Medicines Agency (EMA) on recent, large-scale international efforts to promote knowledge-sharing.
As part of this undertaking, the FDA’s Health Informatics team, the NIH’s National Center for Advancing Translational Sciences (NCATS), and the EMA have collaborated to create the Global Substance Registration System (GSRS). The new system will enable the efficient and accurate exchange of information on the substances in a regulated product, giving users the ability to track a product and its different components as it circulates through the supply chain.
GSRS is groundbreaking because it addresses an unmet need. Consumers, clinicians, and researchers often have no way of telling which ingredients are contained in commercial products that are domestically and foreign-sourced. Certain ingredients are not included in the labeling, and others are labeled ambiguously. Food product labels, for example, often contain general terms like “spices” or “artificial flavoring.”
Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base allows substances to be defined by standardized, scientific descriptions. It classifies substances as chemical, protein, nucleic acid, polymer, structurally diverse, or mixture, as detailed in the International Organization for Standardization’s ISO 11238 and ISO DTS 19844.
By providing a common system with standardized content, GSRS will enable the international monitoring of products and ingredients and help the global community achieve the goal of an accurate substance repository.
FDA Addresses Whole Genome Sequencing Feasibility with MPEDA
The FDA has begun taking the early steps needed to help build the capacity of laboratories maintained by the Government of India’s Marine Products Export Development Authority (MPEDA), under a program funded by the U.S. Congress.
In FY 2021 and FY 2022, the U.S. Congress provided $6 million to develop and implement options for regulating shrimp imports, including shrimp from the three largest shrimp-exporting countries to the United States by volume over the last three calendar years — India, Ecuador, and Indonesia.
For this endeavor, the FDA is using a new form of arrangement with these countries that the FDA calls a Regulatory Partnership, which aims to leverage commodity-specific oversight systems, along with data and information, to strengthen food safety before and at the port of entry.

Staff from MPEDA (left) and the FDA (right) meet to discuss the WGS Feasibility Study.
The FDA would like to build on previous training and data exchange activities with MPEDA by providing Whole Genome Sequencing laboratory equipment, supplies, and support for lab personnel. Enhancing MPEDA’s laboratory capacity for WGS, in particular, is seen by the FDA as one way to more rapidly respond to foodborne disease outbreaks both in India and the United States. A laboratory that could participate in the GenomeTrakr laboratory network by collecting, preparing, sequencing, and uploading information on foodborne isolates from shrimp to the National Center for Biotechnology Information’s (NCBI) public databases will expand the pool of data used by public health agencies to perform surveillance on emerging pathogenic threats and mount appropriate responses to outbreaks. (The India Office’s earlier facilitation of a Confidentiality Commitment with MPEDA in January 2022 provides a framework in which such data and information can be shared.)

MPEDA welcomes Sarah McMullen and Mark Abdoo with a customary token of flowers. The FDA visitors in front of the MPEDA office in Kochi, Kerala.
On September 26, FDA officials, led by Associate Commissioner for Global Policy and Strategy Mark Abdoo, visited the MPEDA lab in the city of Kochi, within the Indian state of Kerala, to consider MPEDA’s roles and responsibilities, the FDA’s approach to WGS, how WGS fits into the Regulatory Partnership Arrangement, and how WGS can be used by India. The FDA team also conducted a feasibility study of the lab and determined that MPEDA was ready to receive WGS equipment. Other FDA participants included the FDA India Office’s Director Sarah McMullen, Deputy Director Natalie Mickelsen, Food Safety Coordinator Pankaja Panda; and a team from the FDA Center for Food Safety and Applied Nutrition, including International Policy Analyst Anthony Rizkalla, Research Microbiologist Ruth Timme, and Research Microbiologist Padmini Ramachandran.
The following day, the FDA team visited Mangala Seafoods in the Aroor suburb of Kochi. The purpose of this seafood factory visit was to visit a sampling location site and learn about sample collection procedures. MPEDA laboratory personnel demonstrated how a shrimp sample is collected and transported to the laboratory for analysis. Mangala Seafoods exports shrimp and squid to the United States.
U.S. Selects Lead Pandemic Accord Negotiator
Secretary of Health and Human Services Xavier Becerra and Secretary of State Antony J. Blinken recently announced the selection of Ambassador Pamela K. Hamamoto as lead U.S. negotiator for the proposed accord on pandemic prevention, preparedness, and response currently being discussed at the World Health Organization (WHO). The announcement confirms the U.S. commitment to take a whole-of-government approach to the negotiating process by establishing a team led by the State Department and HHS, while actively engaging other federal agencies responsible for development, security, economic, and other issues.

The FDA is one of the HHS operating divisions supporting the Department’s efforts, with the Office of Trade, Mutual Recognition, and International Arrangements taking a lead role for the agency.
“As lead U.S. Pandemic Negotiator, Ambassador Hamamoto will assume management and oversight of U.S. engagement in these important discussions, which we believe must yield an accord that effectively strengthens global health collaboration, improves systems for monitoring disease or pandemic outbreaks, bolsters national health security capacities, and enhances equity in pandemic preparedness and responses,” said Becerra and Blinken in a joint statement issued on October 11.
Prior to her selection for this assignment, Hamamoto served as the United States’ permanent representative to the Office of the United Nations and Other International Organizations in Geneva, where she regularly engaged international leaders on a range of global issues, including humanitarian affairs, sustainable development, global health, human rights, peace and security, women’s empowerment, internet governance, and climate change.
Hamamoto was instrumental in the 2014 launch of the Global Health Security Agenda, an international effort that has grown to more than 70 partners focused on strengthening the world’s ability to prevent, detect, and respond to infectious disease threats. She also helped coordinate early responses to the 2015 Ebola outbreak in West Africa, working across regions and institutions to garner support for the three affected countries and facilitate an effective WHO response.
New PAHO Director Elected by Member States
The Pan American Health Organization (PAHO) recently elected Jarbas Barbosa da Silva Jr., M.D., Ph.D., as its new director. Barbosa, a Brazilian national, was elected to the position last September by PAHO member states during the 30th Pan American Sanitary Conference. He will begin his five-year term in February 2023, succeeding Dr. Carissa F. Etienne of Dominica, who has led the organization since 2012.
Courtesy of PAHO.
Currently the assistant director of PAHO, Barbosa has been leading the organization’s efforts to increase equitable access to COVID-19 vaccines and enhance regional capacities to produce medicines and other health technologies.
Barbosa received his medical degree from the Federal University of Pernambuco, Brazil, and specialized in public health and epidemiology at the National School of Public Health at the Oswaldo Cruz Foundation in Rio de Janeiro. He holds a master’s in medical sciences and a doctorate in public health from the University of Campinas in São Paulo. Earlier in his career, he was municipal secretary of health of Olinda, state secretary of health in Pernambuco, and director of the National Center for Epidemiology in Brasília.
He joined PAHO in 2007 as area manager for health surveillance and disease management, coordinating regional activities related to veterinary public health, health analysis and statistics surveillance, and prevention and control of communicable and noncommunicable diseases.
In 2011, Barbosa rejoined Brazil’s Ministry of Health as secretary of health surveillance, and later became secretary of science, technology, and strategic supplies. Prior to his tenure as PAHO’s assistant director, Barbosa was director-president of the Brazilian Health Regulatory Agency (ANVISA) from 2015-2018.
The Pan American Health Organization is the official health organization for the region of the Americas and serves as the regional office for the World Health Organization.
|
|
|
Back to TOP

In celebration of One Health Day this November 3, we’ve included this special section to highlight One Health activities.
How OGPS Contributes Globally through a One Health Lens
Many centuries ago, Tibetan wisdom-keepers recognized that the health of people, animals, and the environment are fundamentally intertwined. Today, that way of thinking is reflected in a concept known as One Health, which recognizes the importance of looking across disciplines and sectors to solve public health challenges. In today’s precarious world, this approach has never been more important. The FDA has a global role to play through the activities of its foreign offices.
Getty Images.
What is the aim of One Health?
One Health acknowledges the interconnectedness of human, animal, plant, and environmental health. This interface becomes more important as humans continue to expand into previously undisturbed lands and habitats, and as international trade and travel increase exposure to pathogen-transmitting vectors and speed the spread of disease outbreaks.
Consequently, a One Health approach is gaining traction for its holistic perspective in addressing the complex health challenges facing our society, such as ecosystem degradation, food system failures, infectious diseases, and antimicrobial resistance. This transformative approach can be embraced to promote food security, health equity, sustainable development, and integrative and innovative approaches to challenges.
Applying the One Health concept to public health replaces piecemeal approaches with comprehensive solutions. This means collaborating across disciplines by bringing together physicians, veterinarians, environmental scientists, social scientists, engineers, public health professionals, regulators, and policymakers to collectively forge a path forward for the best chance of sustainable success.
How is the FDA getting involved?
At the FDA, the Office of Chief Scientist, within the Office of the Commissioner, together with the Director of the Center for Veterinary Medicine, oversee the FDA’s One Health Initiative in collaboration with nine FDA centers and offices. Leveraging science, technology, and innovation, the FDA One Health initiative seeks to: create a multidisciplinary mindset for internal and external FDA stakeholders; provide infrastructure to sustain the effort; engage governmental partners in coordination, communication, outreach, and inclusivity; and reach globally to expand the FDA’s One Health impact.
As we prepare to observe One Health Day on November 3, it is worth recognizing that the FDA’s Office of Global Policy and Strategy (OGPS) — often working with other FDA centers and offices, U.S. and foreign government agencies, and international, regional, and industry organizations — has been influential in shaping a diverse portfolio of global FDA activities that address One Health concerns at fundamental levels.

Large-scale shrimp farms in Latin America. Getty Images.
How do OGPS activities contribute to global One Health?
The OGPS foreign offices in Europe, India, Latin America, and China are involved with many activities that have a One Health component. For the staff, it’s not only about keeping imported food or medical products safe, but about how we, as regulators, approach our relationships with our global counterparts and strive together, mindfully, toward solutions. A small sampling of the broad range of our foreign offices’ endeavors is highlighted below.
-
WGS networks and capabilities. Our foreign offices have been at the forefront of promoting international participation in the GenomeTrakr network, which allows government agencies, laboratories, and academia across the globe to share and compare whole genome sequencing (WGS) data from food and environmental samples. The broader the participation, the better public health agencies can perform surveillance on emerging pathogenic threats and mount an appropriate response to outbreaks.
This August, the Latin America Office facilitated bilateral engagements between the FDA and food agencies in Mexico to increase their participation in the GenomeTrakr network. This is an important step and will allow the agencies to identify and respond to outbreaks faster and with more precision. Similarly, the India Office is currently working to facilitate transfer of WGS instrumentation to India’s Marine Products Export Development Authority (MPEDA) and to provide training so MPEDA can upload the genetic information on isolates to the GenomeTrakr network. This will be the first use of WGS for food safety purposes by a regulatory body in India. [For more, please see our preceding article on the India Office’s WGS activity with MPEDA.]
The Europe Office has led and coordinated several bilateral discussions with key EU entities involved in foodborne pathogen surveillance and foodborne outbreak response, in part to better understand the application of WGS in the EU’s food safety system. Building on these bilateral efforts, the FDA is bringing together experts in food, the environment, and human health for a broader discussion on WGS. The FDA is currently planning a U.S.-EU dialogue involving the U.S. Centers for Disease Control and Prevention and key EU agencies to discuss challenges and opportunities for WGS data sharing and public access through the GenomeTrakr network; the workshop is scheduled for January 12-13, 2023, and will be hosted by the Europe Office.
Looking ahead, the development of stronger WGS networks and surveillance and a focus on supply chain traceability are important next steps to broadening our approach to food safety beyond the walls of the manufacturing facility and preventing disease outbreaks at the source.
-
Produce safety. The Latin America Office has been at the forefront of outreach regarding education on safe produce practices that include awareness and mitigation of disease vectors from wildlife, the surrounding environment, byproducts of nearby human activities, and improper human handling (e.g. unclean hands). Through a cooperative agreement with the Inter-American Institute for Cooperation on Agriculture, the Latin America Office (with the FDA’s Center for Food Safety and Applied Nutrition, or CFSAN) helped facilitate numerous trainings on produce safety aimed at reducing foodborne outbreaks, such as those involving leafy greens or fresh fruit and vegetables from pathogens including salmonella and cyclospora.
-
Seafood safety. The amount of seafood consumed in the United States and throughout the world is growing. With this increased consumption, aquaculture products have become a more important source for people’s nutritional and food security needs. Aquaculture often occurs in or near large bodies of water where the farm may share a contiguous environment with nearby wildlife. The Latin America and India Offices are supporting the FDA’s One Health approach to ensure a safe supply of aquacultured seafood. Together with CFSAN, these offices are helping to provide training for regulators and industry in these regions on Seafood HACCP and good aquaculture practices (GAqP) — which address natural hazards, food safety, and proper use of veterinary drugs, including antimicrobial stewardship.
-
Pandemic readiness and vaccine equity. The India Office supported global pandemic response efforts by conducting inspections at a vaccine manufacturer in India on behalf of the European Medicines Agency (EMA) to facilitate regulatory assessment of vaccines being considered for Emergency Use Listing by the World Health Organization and inclusion in COVAX (short for COVID-19 Vaccines Global Access, a worldwide initiative aimed at equitable access to COVID-19 vaccines). In support of the India Office efforts, the Europe Office facilitated discussions, as needed, through their dedicated FDA Liaison to the EMA.
The Latin America Office provided regional outreach opportunities and worked closely with regulatory partners to share information on the FDA’s regulatory pathways related to emergency response and Emergency Use Authorizations, which was utilized by many Latin American countries to streamline their own respective approval processes of essential medical products and to create regulatory pathways for making decisions in response to public health emergencies.
-
Antimicrobial resistance. Over the past three years, the Europe Office has facilitated discussions between the FDA and the EMA and participated in interagency discussions with the European Commission (EC) while the EU was developing new legislation about the use of antimicrobials. This legislation lists antimicrobials reserved for treating infections in humans only, and therefore these products are not authorized for use on animals (for example in farming). The EU finalized its legislation in July 2022. Although the list itself presents no concerns (i.e., no drugs approved for food animals in the United States are on this EU list) the Europe Office is currently monitoring the adoption of a delegated act (a non-legislative act by the EC to supplement or amend certain non-essential elements of a legislative act) that will clarify the application of specific provisions affecting third countries.
-
Participation in international organizations and events. All of the OGPS foreign offices routinely and extensively attend, help facilitate, or present at meetings, conferences, and events in their respective regions on One Health-related topics; and some staff may even participate in technical working groups along with international regulatory counterparts. This engagement inspires new insights and provides opportunities for the FDA to better understand our global counterparts and stakeholders.
 |
Growing peppers. Getty Images.
Looking Ahead
The COVID-19 pandemic has been an unrelenting reminder that a One Health approach is critical if we want to effectively combat future outbreaks before they turn into a pandemic. Our global world today cannot afford to ignore animal diseases and their risk to public health, as over 60% of emerging infectious diseases globally are zoonotic. Moreover, produce outbreaks and recalls occurring over the last several decades have shown many causes to be of animal origin.
The direct toll on human health is only part of the global effect, which also puts a strain on medical product resources and impacts local and international food supply chains.
As we have recapped, the FDA’s foreign offices remain ever alert to these interconnections and are diligently applying the agency’s broad array of regulatory tools to protect public health.
The Quadripartite Announces One Health Joint Plan of Action
On October 17, four international agencies — the Food and Agriculture Organization of the United Nations (FAO), the United Nations Environment Programme (UNEP), the World Health Organization (WHO), and the World Organisation for Animal Health (WOAH, founded as OIE) — announced the launch of their first joint plan on One Health.
Their cooperation on One Health was formalized in March 2022 when the four signed a Memorandum of Understanding for joint One Health activities, forming the Quadripartite Collaboration for One Health. The MOU provides a legal and formal framework for the agencies to use a more integrated approach when confronting challenges at the human, animal, plant, and ecosystem interface.
The Quadripartite agencies’ One Health Joint Plan of Action “aims to create a framework to integrate systems and capacity so that we can collectively better prevent, predict, detect, and respond to health threats,” the WHO said in a press release. “Ultimately, this initiative seeks to improve the health of humans, animals, plants, and the environment, while contributing to sustainable development.”

The plan extends for five years, through 2026, and focuses on building the agencies’ strengths across the most critical of global One Health concerns:
- One Health capacities for health systems.
- Emerging and re-emerging zoonotic epidemics.
- Endemic zoonotic, neglected tropical, and vector-borne diseases.
- Food safety risks.
- Antimicrobial resistance and the environment.
The four collaborating agencies seek to provide a framework for adoption of a One Health approach to coordinated, multisectoral action and partnership that can be embraced by all organizational levels — governments and international agencies, industry organizations and professional associations, and academia and research institutions.
According to the WHO, the plan sets out operational objectives to include “upstream policy and legislative advice and technical assistance to help set national targets and priorities” and to promote “multinational, multisector, multidisciplinary collaboration, learning and exchange of knowledge, solutions and technologies.” At the core of the plan are the values of cooperation and shared responsibility; a set of values that the world is now at a crossroads to address.
The World Bank Calls for Moving Away from Pandemic Containment to Pandemic Prevention
In advance of One Health Day on November 3, the World Bank has issued a report urging policymakers, governments and the international community to invest in pandemic prevention and move away from what it calls “the business-as-usual approach” based on containment and control after a disease has emerged. The report estimates that prevention guided by a One Health approach — sustainably balancing and optimizing the health of people, animals, and ecosystems — would cost anywhere from $10.3 billion to $11.5 billion per year. In contrast, the G20 Joint Finance and Health Taskforce has estimated that it costs about $30.1 billion per year to manage pandemics. Moreover, actions to prevent disease outbreaks carry an estimated rate of return of up to 86% and most of these actions will result in significant co-benefits, the World Bank says.
“Prevention is better than cure. COVID-19 has shown that a pandemic risk anywhere becomes a pandemic risk everywhere. The economic case for One Health is powerful — the cost of prevention is extremely modest compared to the cost of managing and responding to pandemics,” said Mari Pangestu, World Bank Managing Director of Development Policy and Partnerships.
To make its point, the report, “Putting Pandemics Behind Us: Investing in One Health to Reduce Risks of Emerging Infectious Diseases,” includes case studies of Liberia, Vietnam, and the Assam State in India.
Cooperating for a healthier future. Getty Images.
|
|
|
Back to TOP

Promotion

Marcus Ray
Marcus Ray is the new medical products supervisory consumer safety officer in the China Office as of September 26. He replaces Jonathan Chapman, who recently moved to a policy position in the office. Ray has been a drug investigator for the China Office since 2020.
|
Relocation
Bruce Ross, former director of the Office of Global Operations, has now begun serving as an international policy analyst for foods for the Latin America Office, at post in Santiago, Chile.
Detailees
We give our gratitude and goodbyes to Lisa Lopez for her time as Acting Deputy Director of the China Office from July through September.
Sandy Kweder’s Retiring — Yes — But
The FDA without Sandy Kweder? Say it’s not so!
Over the course of 25 years Sandy Kweder, M.D., has been the consummate Deputy Director, with a demonstrated history of public service focused on pharmaceutical development and regulation across the medical product life cycle. She first joined the FDA in the offices of antiviral drugs and postmarketing safety before pulling up roots to do a fellowship at Brown University in medical complications of pregnancy. Sandy then returned to the FDA for leadership roles in the Center for Drug Evaluation and Research (CDER), and ultimately as Deputy Director in CDER’s Office of New Drugs that oversees the development and review of all innovator drugs. She then took on the challenge of Deputy Director in the FDA’s Europe Office and liaison to the European Medicines Agency, where she brought her experience to bear internationally, building collaborations and global alignment on regulatory science and policy.

One big reason she’s flourished in these roles is that Sandy has a rare gift among doctors and other scientific experts: She seeks true communication with others, finding common ground, and is able to cut through jargon to communicate about difficult and complicated topics in crisp, compelling, understandable terms. That’s why it was Sandy who was tapped to lead the overhaul of FDA’s Pregnancy and Lactation Labeling Rule in 1998, and to appear before a congressional committee to discuss Merck’s voluntary recall of the nonsteroidal anti-inflammatory drug Vioxx in 2004, one of most widely used drugs ever to be withdrawn from the market. And why it was that Sandy served as the FDA spokesperson during a two-day public meeting in 2009 on a common and commonly prescribed drug — acetaminophen (Tylenol anyone?) — to explain the risks of mixing over-the-counter drugs containing acetaminophen or of downing the pain killer with an alcoholic drink or two. There are many more examples. No wonder the Commissioned Corps of the U.S. Public Health Service awarded Sandy the rank of Rear Admiral in 2007.
OGPS has its own confirmation of Sandy’s communication skills. Just take a look at the new OGPS web page on working abroad. You’ll see Sandy’s face gracing a crisp two-minute video on the benefits of working abroad.
Sandy also has academic chops. Before leaving for her European assignment, she maintained a faculty appointment, teaching medical students at the Uniformed Services University of Health Sciences and serving as an internal medicine specialist attending at Walter Reed Military Medical Center. In keeping with Sandy's drive to communicate, in the Europe Office, her co-authored paper “A Comparison of EMA and FDA Decisions for New Drug Marketing Applications 2014–2016: Concordance, Discordance, and Why” won an award from the journal Clinical Pharmacology and Therapeutics, for best representing the type of research articles that the journal strives to publish, those that are “timely, influential and carefully researched.” It was the first of a number of articles published, including a newly published one on the work of FDA and EMA partnerships: "Considering Global Development? Insights from Applications for FDA Breakthrough Therapy and EMA PRIME Designations."

On top of the world: hiking the Dolomite Alps in Italy.
When you talk to Sandy, she says what's always motivated her at the FDA is the value of bringing experts together to build bridges across the agency. “I love the camaraderie of our divisions, offices, and especially our centers, but we need to remember that science and clinical medicine don’t understand or care about organograms [org charts to the Europeans],” Sandy says. “We are fortunate to have so many bright scientific minds here at the agency. Our challenge is to facilitate and encourage working together, synergizing to bring science to life, and public health actions that positively affect the lives of people we may never meet. This has become more difficult since COVID-19, but we should not be deterred as our biggest successes arise when we do.”
The FDA leadership has certainly paid attention to Sandy’s skills and accomplishments. This year she was one of only a handful of staffers to receive the prestigious FDA Award of Merit for her international work.
Other ways to describe Sandy? Surely, she must be the original Energizer Bunny with loads of energy she pours into such endeavors as bagging Munros in Scotland, hiking the Dolomites, cycling the Netherlands, reviving a street garden in Amsterdam, learning to row a scull on the Amstel River, and coming home from Europe several times to join her sisters in baking over 1,000 Italian cookies for family weddings. Most recently some of us have watched in wonder here at White Oak as she hoists those hand weights in the gym, which she says is to set a good example for the Masters Swim Team she now coaches on weekends!

Sandy enjoys flowers, gardening, and being outside with nature!
Joie de vivre? Certainly. Or perhaps you could say she is simply game to take on anything. EO Director Ritu Nalubola recalls her first encounter with Sandy, at an international meeting, when after an impromptu invitation, Sandy joined Irish counterparts on stage in an Irish dance. “She hasn’t failed to surprise me with her many talents on other occasions since then,” Ritu said. In commenting on Sandy’s departure from Amsterdam, earlier this year, Ritu noted: “I will miss her sound counsel and guidance and, most of all, the lighthearted spirit, enthusiasm, and humor she brings to every conversation.”
The FDA without Sandy Kweder? Say it’s not so. But alas, for us, it is. Sandy retires from federal service on November 20. “No. I do not have a job yet and that suits me quite well for now,” she said in a brief note to OGPS staff. For now, she says she just wants to be the woman in the neighborhood with the light on. Knowing Sandy, she will surely be active. In fact, we can’t wait to hear what she plans to do next.
|
|
|
Back to TOP

Recent communications from OGPS to our international stakeholders (list does not include twice-weekly FDA Roundup summaries), September 21 through November 2.
|
|
|
Back to TOP

|
November 3
|
One Health Day
|
|
November 3, 10, 21
|
The Fogarty International Center at NIH presents a three-part webinar series on Global Food and Nutrition Insecurity
|
|
December 1
|
World AIDS Day
|
|
|
|
Read thought-provoking pieces covering international topics in
From A Global Perspective.
|
|
|
Please follow @FDA_Global.
|
|
|
Subscribe to Global Update
Don't miss out on future issues of Global Update or other international news from OGPS. To sign up, click "Manage Subscriptions" in the below footer then follow the prompts and select the "International Programs" box.
|
|
|
|
|