|

Discover how scientists and engineers in the Office of Science and Engineering Laboratories (OSEL) at the U.S. Food and Drug Administration’s (FDA) Center for Devices and Radiological Health (CDRH) promote innovation for the development of new lifesaving medical devices.
2022 Regulatory Science Tools Webinar Series: Part I
The National Heart, Lung, and Blood Institute's (NHLBI’s) Small Business Program is partnering with CDRH for a free, three-part webinar series examining medical device innovation with regulatory science tools.
Who should attend:
- Entrepreneurs
- Small business innovators who are interested in using regulatory science tools for emerging technologies
Hear from distinguished leaders in the small business and med-tech innovation space including NHLBI, CDRH, and the Small business Education and Entrepreneurial Development (SEED) office.
Date: January 19, 2022
Time: 1:00PM EST - 2:00PM EST
OSEL Program Highlights

Job Opportunities
Interested in a science or engineering career at the heart of public health and medical device innovation? CDRH’s OSEL has several openings for candidates with a strong background in a variety of scientific disciplines, including laboratory research policy or consulting support for reviews of new medical devices, and analysis of device failures.
Read more and apply today at Job Opportunities at OSEL.
|
Medical Device Material Safety Summaries: 5 New ECRI Reports
As a part of the FDA’s ongoing commitment to promote the safety of medical devices, we have partnered with ECRI (originally founded as Emergency Care Research Institute) to publish safety summaries for materials that are commonly used in implantable medical devices and the effects of those materials on patients over time.
Five new safety summaries are now available.
-
PET (polyethylene terephthalate), used broadly
-
PEG (polyethylene glycol), used broadly and as a coating for stents and catheters
-
Silver, used as an antimicrobial agent
-
Acrylic acid derivatives, which includes di-, tri- and glycerol methacrylates, often used in dental resins
-
Polyhydroxyacids, including PLA, PGA, and other blends and copolymers, the most common class of bioresorbable polymers
We will continue to release new safety summaries as they become available. Read more about the Medical Device Material Safety Summaries: ECRI Reports.
3D Printing and Additive Manufacturing as a Solution to Supply Chain Gaps
A report published in the New England Journal of Medicine’s (NEJM) Catalyst (Innovations in Health Care) on November 10, 2021, describes the conception, implementation, and effectiveness of the Covid 3D TRUST initiative and highlights two keys to its successful rapid response: (1) trust among our respective groups and the diverse community of stakeholders and contributors and (2) value added for designers, manufacturers, and end-users of 3D-printed PPE and medical supplies.
Read more in Trust in the Time of Covid-19: 3D Printing and Additive Manufacturing (3DP/AM) as a Solution to Supply Chain Gaps | Catalyst (nejm.org)
Virtual MRI Safety Evaluations of Medical Devices
- Is a non-clinical assessment model (NAM), that uses computational modeling and simulation to predict the interactions between medical device implants and electromagnetic fields in the magnetic resonance (MR) environment.
- Can reliably predict the temperature rise caused by radiofrequency (RF) heating near electrically passive, fully implanted, medical devices such as orthopedic joint prostheses and cardiovascular stents.
- Has the potential to more efficiently generate results compared with bench testing certain medical devices.
Read more about Medical Device Development Tools (MDDTs).
Assessing Dosing Errors in Legacy and Low Dose Tip ENFit Syringes
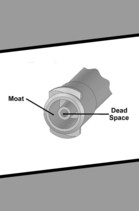
The Journal of Clinical Pharmacy and Therapeutics published the following article: Assessing Dosing Errors in Legacy and Low Dose Tip ENFit Syringes on October 29, 2021.
In the study, a research team at the FDA assessed the risk of medication overdose from the moat area of ENFit low dose top (LDT) syringes by using computer-aided modeling to analyze the maximum possible dosing errors in 0.5 mL and 1.0 mL ENFit LDT syringes and traditional syringes under various clinical use conditions, including varied filling and administration methods.
Study results showed dosing errors are possible for both traditional syringes and the ENFit LDT syringes, depending on the filling and administration methods used. The FDA believes that dose accuracy using ENFit LDT syringes can be optimized when users follow the recommendations in the Potential for Medication Overdose with ENFit Low Dose Tip Syringe: FDA Safety Communication.
|
 |
|
"FDA provides a unique opportunity to work together with experts across a wide range of specialties with the unifying goal of improving public health.”
Ryan Beams, PhD, Physicist | FDA
|
Catalog of Regulatory Science Tools to Help Assess New Medical Devices
Contact OSEL
If you have questions about OSEL, email OSEL_CDRH@fda.hhs.gov.
You can also subscribe to CDRH Science for regular email updates on CDRH’s scientific topics and OSEL.
|