|
Coronavirus Disease 2019 (COVID-19) updates
Actions by FDA in our ongoing response to the COVID-19 pandemic since our last MCMi email update include:
Bookmark www.fda.gov/coronavirus for the latest.
|
|
|
COVID-19 vaccine updates
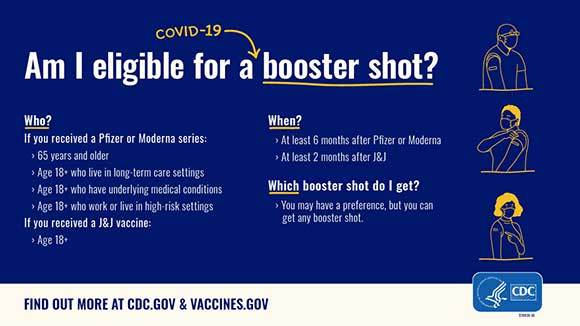 ICYMI - FDA takes additional actions on the use of a booster dose for COVID-19 vaccines
On October 21, 2021, the FDA took action to expand the use of a booster dose for COVID-19 vaccines in eligible populations. The agency amended the Emergency Use Authorizations (EUAs) for COVID-19 vaccines to allow for the use of a single booster dose.
Updated translations of the fact sheets for recipients and caregivers will be posted on each of the vaccine information pages from FDA as they become available:
Additional information about FDA's review is also available at the pages above, and on FDA's EUA web page. Also see, from CDC: CDC Expands Eligibility for COVID-19 Booster Shots.
|
|
Emergency Use Authorization (EUA) updates

FDA authorizes additional COVID-19 OTC antigen tests, updates EUA templates to further support OTC test availability
On October 25, 2021, FDA authorized the 10th over-the-counter (OTC) COVID-19 test, reissued the EUA for another OTC COVID-19 test, and updated the EUA templates for test developers seeking an EUA for OTC tests. The FDA:
-
Issued an EUA (PDF) for the Celltrion Diatrust COVID-19 Ag Home Test, an OTC COVID-19 diagnostic antigen test. The test can be used by people with COVID-19 symptoms as a single test without a health care provider prescription, or by people with or without symptoms for serial testing, which means the test is performed two times over three days.
-
Reissued the EUA (PDF) for the Quidel QuickVue At-Home OTC COVID-19 Test to authorize use by people with COVID-19 symptoms as a single test without a health care provider prescription. The test is already authorized for use by people with or without symptoms as a serial test and for people with COVID-19 symptoms as a single test with a health care provider prescription.
- Updated the test labeling recommendations for test instructions and product labeling in the EUA templates for test developers to support authorization of more COVID-19 tests for use without a health care provider prescription. The updated templates also include flexible study recommendations about how to demonstrate that different types and ages of consumers can use the test appropriately.
Shipment of bamlanivimab/etesevimab resumes to Hawaii
Based on FDA’s evaluation of the most recently available SARS-CoV-2 variant frequency data, we are informing you that the combined frequency of variants resistant to bamlanivimab and etesevimab administered together is now less than 5% in all U.S. states, territories, and jurisdictions, including Hawaii. Bamlanivimab and etesevimab, administered together, can be used in all U.S. states, territories, and jurisdictions consistent with the terms and conditions of authorization for EUA 94 (PDF). As such, ASPR will resume the distribution of bamlanivimab and etesevimab together and etesevimab alone (to pair with existing supply of bamlanivimab at a facility for use under EUA 094) to Hawaii and will continue distribution to all other U.S. states, territories, and jurisdictions (PDF). (October 21, 2021)
In vitro diagnostic (test) EUAs
As of October 26, 2021, 420 tests and sample collection devices are authorized by FDA under EUAs. These include 292 molecular tests and sample collection devices, 90 antibody and other immune response tests, and 38 antigen tests. There are 66 molecular authorizations and 1 antibody authorization that can be used with home-collected samples. There is 1 molecular prescription at-home test, 3 antigen prescription at-home tests, 9 antigen over-the-counter (OTC) at-home tests, and 2 molecular OTC at-home tests. FDA has authorized 16 antigen tests and 8 molecular tests for serial screening programs. The FDA has also authorized 663 revisions to test EUA authorizations. Also see: Coronavirus Testing Basics
|
|
|
Events
-
New! November 8 - 9, 2021: 13th Annual Sentinel Initiative Public Workshop - This virtual public workshop will highlight milestones and strategic initiatives underway to enhance and build a more robust Sentinel Initiative. Stakeholders will discuss opportunities to utilize Sentinel’s existing data, infrastructure, and technology.
-
New! November 9, 2021: FDA CERSI Lecture on Long COVID: Risk factors, Symptomology and Patient Reported Outcomes Captured Through a Novel Digital Platform, presented by by Dr. Erica Spatz & Dr. Kelli O’Laughlin, 3:00 - 4:00 p.m. ET (virtual)
-
November 16, 2021: Enhanced Drug Distribution Security at the Package Level Under the Drug Supply Chain Security Act (DSCSA) (virtual) - To provide members of the pharmaceutical distribution supply chain and other interested stakeholders an opportunity to discuss enhanced drug distribution security requirements of the DSCSA related to system attributes necessary to enable secure tracing of product at the package level.
-
November 17, 2021: Virtual Town Hall Series - Coronavirus (COVID-19) Test Development and Validation, 12:15 p.m. - 1:15 p.m. ET, hosted by FDA's Center for Devices and Radiological Health (CDRH). FDA will host additional events in this series December 1 and December 15, 2021.
-
November 30, 2021: Antimicrobial Drugs Advisory Committee (AMDAC) meeting (webcast) - To discuss Merck and Ridgeback’s request for an EUA for molnupiravir, an investigational antiviral drug to treat COVID-19. The committee will discuss the available data supporting the use of molnupiravir to treat mild-to-moderate coronavirus disease 2019 (COVID-19) in adults who have tested positive for COVID-19, and who are at high risk for progression to severe COVID-19, including hospitalization or death.
-
December 7-8, 2021: Clinical Investigator Training Course (CITC) Update (virtual), including a review of the development and emergency use authorization of medical products for the prevention and treatment of COVID-19, and investigator responsibilities during the pandemic.
|
|
Information for industry and health care providers
Shelf life extension of etesevimab under the EUA for bamlanivimab and etesevimab administered together
FDA and HHS ASPR announced the authorization of an extension to the shelf-life from 12 months to 18 months for the refrigerated Eli Lilly monoclonal antibody, etesevimab, which is currently authorized for emergency use only when administered together with bamlanivimab. As a result of this extension, unopened vials of etesevimab, injection, 700 mg/20 mL, should be stored under refrigerated temperature at 2°C to 8°C (36°F to 46°F), and may be stored for an additional 6 months from the labeled date of expiry (See Table 1 here). FDA granted this extension following a thorough review of data submitted by Eli Lilly. This extension applies to all unopened vials of etesevimab that have been held in accordance with storage conditions detailed in the authorized Fact Sheet for Health Care Providers (PDF) and the Letter of Authorization (PDF) for Emergency Use Authorization (EUA) 094 for bamlanivimab and etesevimab, administered together. (October 22, 2021)
FDA is committed to providing timely recommendations, regulatory information, guidance, and technical assistance necessary to support rapid COVID-19 response efforts. FDA has issued more than 75 COVID-19-related guidances to date.
|
|
In case you missed it

It's a good time to get your flu vaccine
Influenza (flu) viruses typically spread in fall and winter, with activity peaking between December and February. Getting vaccinated now can lower your chances of getting the flu.
|

Why you should not use ivermectin to treat or prevent COVID-19
Some people are taking ivermectin, a drug often prescribed for animals, to try to prevent or treat COVID-19. FDA has not approved or authorized ivermectin for this use, and it can be dangerous for people. Information is also available in six additional languages.
|

List of hand sanitizers consumers should not use
Some hand sanitizers have been recalled and there are more than 250 hand sanitizers the FDA recommends you stop using right away. Bookmark www.fda.gov/handsanitizerlist for the latest, and use our step-by-step search guide to find out if your product is on the list.
|

Learn more about COVID-19 vaccines
Getting vaccinated to prevent COVID-19 will help protect you from COVID-19, and it may also protect the people around you. Learn about the evidence supporting FDA approval of Comirnaty and FDA emergency use authorization of COVID-19 vaccines.
|
Did someone forward you this email? Subscribe.
(Select Emergency Preparedness and Response,
FDA Medical Countermeasures Initiative (MCMi) News)
|
|
|
|
|