Coronavirus Disease 2019 (COVID-19) updates
Actions by the FDA in our ongoing response to the COVID-19 pandemic since our last email update include:
- September 17, 2021: Potential for False Positive Results with Abbott Molecular Inc. Alinity m SARS-CoV-2 AMP and Alinity m Resp-4-Plex AMP Kits - Letter to Clinical Laboratory Staff and Health Care Providers
- September 17, 2021: COVID-19 Update including a revised guidance to help expand the availability of face masks, barrier face coverings, and face shields for the general public, including health care personnel, as well as surgical masks and particulate filtering facepiece respirators (FFRs) (including N95 respirators) for health care personnel for the duration of the COVID-19 public health emergency
- September 17, 2021: The Vaccines and Related Biological Products Advisory Committee met in open session to discuss the matter of additional doses of COVID-19 vaccines and specifically to discuss the Pfizer-BioNTech supplemental Biologics License Application for administration of a third (“booster”) dose of Comirnaty (COVID-19 Vaccine, mRNA) in individuals 16 years of age and older.
- September 16, 2021: FDA authorizes bamlanivimab and etesevimab monoclonal antibody therapy for post-exposure prophylaxis (prevention) for COVID-19
- September 10, 2021: COVID-19 Update including approval of an abbreviated new drug application indicated for the sedation of initially intubated and mechanically ventilated patients during treatment in an intensive care setting
- September 10, 2021: FDA Will Follow The Science On COVID-19 Vaccines For Young Children
Bookmark www.fda.gov/coronavirus for the latest.
|
 COVID-19 vaccine updates
FDA will follow the science on COVID-19 vaccines for young children
We know there have been questions and public commentary on the process surrounding vaccines for young children, so we think it’s important to share information about the process and the necessary considerations involved to provide greater clarity to the public about this effort. Learn about the steps FDA will take to ensure the safety and efficacy of COVID-19 vaccines for young children.
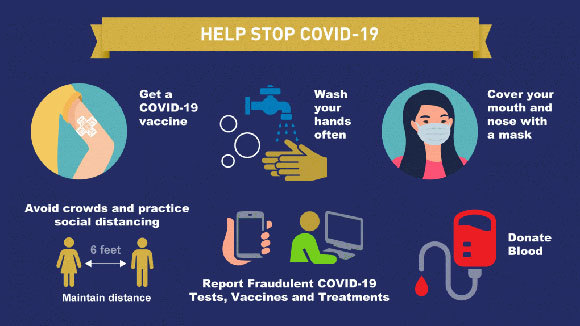 Help stop the spread of coronavirus and protect your family
The COVID-19 pandemic demands that we remain vigilant in our daily lives as we return to everyday activities. We can each take some simple steps to protect ourselves, our families, and our communities.
The steps are:
Get a COVID-19 vaccine.
Wash your hands often with plain soap and water.
Cover your mouth and nose with a mask when around others.
Avoid crowds and practice social distancing (stay at least 6 feet apart from others).
Upcoming events
-
New! September 28, 2021: Public Workshop on COVID-19 Lessons Learned: Clinical Evaluation of Therapeutics, 1:00 - 5:30 p.m. ET, virtual - To share lessons learned from the Federal COVID-19 Response or Countermeasures Acceleration Groups (formerly known as Operation Warp Speed) to bring relevant therapeutics to use in a public health emergency. More info, including agenda and registration available from Reagan-Udall Foundation for the FDA.
-
September 30, 2021: The Vaccines and Related Biological Products Advisory Committee will meet in open session (8:30 a.m. - 3:40 p.m. ET, virtual) to hear an overview of the research programs in the Laboratory of Bacterial Polysaccharides, Division of Bacterial, Parasitic, and Allergenic Products, Office of Vaccines Research and Review, and the Center for Biologics Evaluation and Research (CBER), and to discuss and make recommendations on the selection of strains to be included in the influenza virus vaccines for the 2021 to 2022 southern hemisphere influenza season.
-
October 4-6, 2021: 11th Annual Global Summit on Regulatory Science, hosted by FDA's National Center for Toxicological Research - This year's theme is Regulatory Sciences for Food/Drug Safety with Real-World Data & Artificial Intelligence (AI).
-
October 5, 2021: Enhanced Drug Distribution Security in 2023 Under the DSCSA webinar, 1:00 - 2:00 p.m. ET - To discuss enhanced drug distribution security requirements that go into effect in 2023 under the Drug Supply Chain Security Act (DSCSA), including how enhanced drug distribution security will help protect patients from exposure to drugs that may be counterfeit, stolen, contaminated, or otherwise harmful.
In case you missed it
Why you should not use ivermectin to treat or prevent COVID-19
Some people are taking ivermectin, a drug often prescribed for animals, to try to prevent or treat COVID-19. FDA has not approved or authorized ivermectin for this use, and it can be dangerous for people. Information is also available in six additional languages.
|
|
 |
List of hand sanitizers consumers should not use
Some hand sanitizers have been recalled and there are more than 250 hand sanitizers the FDA recommends you stop using right away. Bookmark www.fda.gov/handsanitizerlist for the latest, and use our step-by-step search guide to find out if your product is on the list.
|
|
 |
Learn more about COVID-19 vaccines
Getting vaccinated to prevent COVID-19 will help protect you from COVID-19, and it may also protect the people around you. Learn about the evidence supporting FDA approval of Comirnaty and FDA emergency use authorization of COVID-19 vaccines.
|
|
 |
Find additional coronavirus disease 2019 (COVID-19) updates on our website and in the Medical Countermeasures Initiative newsletter.
Visit the FDA OMHHE website and follow us on Twitter at @FDAHealthEquity
"Creating a world where health equity is a reality for all." www.fda.gov/healthequity

|