 |
|
Update from the CQC
In July we published our 2017 Controlled Drugs Annual Report. In the report we make four recommendations;
1. Prescribers should ask patients about their existing prescriptions and current medicines when prescribing controlled drugs. Where possible, prescribers should also inform the patient’s GP to make them aware of treatment to minimise the risk of overprescribing that could lead to harm.
2. Commissioners of health and care services should include the governance and reporting of concerns around controlled drugs as part of the commissioning and contracting arrangements so that these are not overlooked.
3. Healthcare professionals should keep their personal identification badges and passwords secure and report any losses as soon as possible to enable organisations to take the necessary action.
4. Health and care staff should consider regular monitoring and auditing arrangements for controlled drugs in the lower schedules, such as Schedules 4 and 5, to identify and take swift action on diversion.
Also for the first time, the report details some of our findings on inspection together with our expectations and highlights some of the key concerns raised at CD LINs. You can find the full report along with the stakeholder annex on our website
|
Use of cannabis for medicinal purposes
Recent cases have shown that the government needs to look more
closely at the possible use of cannabis for medicinal purposes in the UK.
The Chief Medical Officer for England (CMO) has reviewed the
evidence and concluded that there is evidence of medicinal benefit. Following
this advice, the Advisory Council on the Misuse of Drugs (ACMD) considered
how best to balance the potential risk of harm and diversion of cannabis and
cannabis-related products. The ACMD will also provide a long-term
review by July 2019.
Following
this advice, the Home Secretary confirmed that cannabis-derived medicinal
products will be rescheduled.
The
Department of Health and Social Care and Medicines and Healthcare products
Regulatory Agency are now developing a clear definition of what constitutes a
cannabis-derived medicinal product so that cannabis-based products can be
rescheduled and prescribed. Only products meeting this definition will be
rescheduled. Other forms of cannabis will be kept under strict controls
and will not be available on prescription. More details about this
definition and the kinds of products that will be covered in the due course.
In the interim a panel of clinical experts is advising ministers
on licensing applications ensuring decisions are based firmly upon medical
evidence and are taken as swiftly as possible.
The review does not propose to change the status of cannabis as
a class B drug under the Misuse of Drugs Act 1971. The penalties for
unauthorised supply, and possession and production will remain
the same.
Links to:
ACMD Recommendation
(Short-term Review) https://www.gov.uk/government/publications/advice-on-scheduling-of-cannabis-based-medicinal-products
Re- Scheduling of Pregabalin and gabapentin
Home Office are currently working on the final response to the consultation. The aim is to implement changes to the Misuse of Drugs Regulations by early next year. We provide further updates in future newsletters.
Safe Custody Regulations
Following the preliminary questionnaire, Home Office planned to hold a full consultation but this has been put on hold due to capacity constraints in light of recent cases involving medicinal cannabis. The Home Office will keep under review the priority to see if it can be possible to move forward.
 |
|
Update of the Controlled Drugs (Supervision of Management and Use) Regulations 2018
The Department of health and Social Care (DHSC) is committed to reviewing the Controlled Drugs (Supervision of Management and Use) Regulations 2013. An on-line questionnaire ran from 23 April - 21 May asked a wide range of stakeholders for their views on the regulations. Response to the questionnaire were overwhelming in support that the regulations be maintained/replaced. The DHSC is currently analysing responses and will aim to publish a report by the end of the summer, with the recommendation to replace the current regulations and updating where necessary.
Should ministers accept the recommendation to replace the regulations, legislation will be drafted and consulted on with a view to bringing the new regulations into force before the current regulations cease to have effect on 31 March 2020.
|
News from the Patient Safety Sub-Group
Take care when prescribing and administering opioids
We have been made aware there have been a number of serious incidents involving the inappropriate prescribing and administration of opioids where patients have required emergency naloxone reversal. It is important to think carefully about all factors when prescribing opioids in particular:
- Consider patient parameters - age, renal function, weight etc.
- Confirm any recent opioid dose, formulation and frequency of administration, including the use of breakthrough doses
- Ensure dose titration is appropriate and safe
- Check other medicines that have been prescribed for the patient
- Ensure the prescription is clear including which formulation is to be used
- Opioid patches - ensure only the new patch(es) is/are in use. Remove old patch and dispose of it safely
- Where required, continuous infusion of naloxone may be required after initial reversal
If you are not sure, seek help - ask a senior colleague, pain team or pharmacist.
Availability of diamorphine (5mg and 10mg)
In the last edition of the newsletter we informed you of a manufacturing issue with one of the suppliers of diamorphine 5mg and 10mg injection.
Department of Health and Social Care (DHSC) and NHS England (NHSE) have continued to work with Accord and their supplier in Germany and the manufacture of diamorphine 5mg and 10mg injection will soon resume with an anticipated resupply date of beginning of September 2018. DHSC and NHSE have also been working with the remaining supplier, Wockhardt, to support the supply issue. Wockhardt have been able to increase their production of diamorphine 5mg and 10mg but are unable to support the entire market in July and August.
Lessons learned from the death of a child: Safe handling of transdermal patches
A child in Cornwall sadly died due to a fentanyl patch belonging to the parent attaching to the child’s skin. This is a sobering reminder of the importance of patients keeping their medicines out of reach and sight of children. We would like to share some of the specific learning that has come to light as a result of this case:
- Please read the MHRA alert on this issue https://www.gov.uk/drug-safety-update/transdermal-fentanyl-patches-reminder-of-potential-for-life-threatening-harm-from-accidental-exposure-particularly-in-children. This includes a letter for patients and care givers.
- Review the route of medication prescribed when performing a medication review. If patches are prescribed, is it still the safest and most effective way to administer the medication?
- Note the potency of fentanyl patches – a 25 microgram/hour patch is equivalent to an oral morphine dose of 60-90mg in 24 hours.
- Patients who are prescribed medication in patches should be informed that co-sleeping with their children is especially risky
- Patients should dispose of patches where children cannot access them (e.g. putting them in a bin that a child can easily access the contents of is risky)
- Patients should never share their prescribed medication with anyone else
Following an inquest, in June 2018, the BBC news channel ran a story in relation to a baby who died when a pain-relief patch became attached to the skin. The news article can be found on the BBC's website here.
This article was kindly shared with us by the NHS England South West Controlled Drugs Team
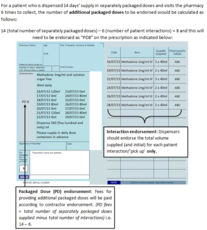
Timely reporting of fraudulent/forged prescriptions
Please ensure that the reporting of fraudulent prescriptions is
timely. This follows a number of incidents reported to NHS England CDAO teams
where fraudulent prescriptions are being presented in pharmacies. Where NHS
England receive notification that lost or stolen prescription forms are in
circulation they issues an alert pharmacies in the locality by e-mail. If you believe
that you are not on their circulation list (because you have never received
any), please contact your local office and inform them of your location and work
e-mail address. Please also ensure that you know how to access your pharmacy’s
NHSnet e-mail address.
This article was kindly shared by the Greater Manchester
Combined Authority - NHS in Greater Manchester Controlled Drugs Team
 |
|
FP10MDA Prescriptions
Further to the article in the last edition, the Prescribing sub-group has been alerted that there has been some prescribing if items not allowed on FP10MDA prescriptions. In England only the following can be supplied in instalments against a FP10MDA prescription:
- Schedule 2 Controlled Drugs
- Buprenorphine (subutex, espranor)
- Bupernorphine/naloxone (suboxone)
- Diazepam
- Diamorphine - requires a specific licence.
Could we please take this opportunity to remind you that if you are using FP10MDA prescriptions outside of the above recommendation please see seek assistance from your local teams include the NHS England CDAO Team.
This article was from NHS England Central Midland CDAO Team.
|
News from the Vigilance Sub-Group
Controlled Drugs Licences
We have been alerted that some NHS providers are unaware of the licenses they need to have in place in order to supply controlled drugs to external providers. This has been a particular issue where the commissioners of substance misuse services stipulate from where the controlled drugs must be procured as part of Health and Justice contracts. NHS providers should check whether they need one or more of the following in order to supply external providers:
1. Home Office CD possession licence
2. Home Office CD supply licence
3. Wholesale Dealers licence
This issue was kindly shared with us by Mohammed Fessal, Chief Pharmacist at Change, Grow Live (CGL)
|