 Latest news from CQC
Controlled Drugs Annual Report 2016
Our update for 2016 highlights CQC’s activity in relation to our
controlled drug responsibilities, including our role with controlled drug local
intelligence networks (CDLINs), the National Group on Controlled Drugs and
the Cross-Border Group for safer management of controlled drugs.
It sets out key changes to legislation, provides an overview of
prescribing data and prescribing trends for controlled drugs across England in
the primary care sector and we make three recommendations to strengthen
existing arrangements.
The annex reports on
activity in 2016 from partner organisations of the controlled drugs national
group and cross-border group.
|
News from the Policy Sub-Group
Human Fertilisation and Embryology Authority
(HFEA) and CDAOs
There are a number of organisations fitting the CDAO
requirements that are registered with the Human Fertilisation and Embryology
Authority (HFEA) and not with the CQC. CQC and HFEA are currently working
together to develop a parallel HFEA CDAO register and we will provide an update
in a future edition.
Controlled Drugs (Supervision of Management and
Use) Regulations 2013
The Department of Health are reviewing the Controlled Drugs
(Supervision of Management and Use) Regulations 2013. An initial meeting was held
during the autumn of 2016 to consider the effectiveness of the regulations and
potential areas to include as part of the review. The Department is in the
processes of planning the next stages of the review.
The review of the Safe Custody Regulations 1973
The Home Office held the first round of meetings to review the
1973 Regulations in early 2017. Meetings have been held with representatives
from both the healthcare and non-healthcare (i.e. museums and universities)
sectors. A set of targeted meetings took place in August 2017 to drill down
into some of the specific issues such as new technology and exemption
certificates.
Diversion from the legal supply chain
MHRA's on-going investigation into diversion of “z drugs
and diazepam ” has now identified around 130 million tablets that have been
diverted from the UK authorised supply chain to the criminal market. Around 40
businesses (wholesale suppliers and pharmacies) have been identified as being
involved and a number of arrests have been made.
Launch of new reporting site for harms
associated with illicit drugs
In March 2017, Public Health England and MHRA launched a new
national system for reporting the effects of new psychoactive substances (NPS)
and other illicit drugs, in a similar way to how adverse effects of
pharmaceutical drugs are reported using the MHRA’s Yellow Card Scheme. This
system is being piloted in order to collect more information about the harms of
new drugs, many of which are poorly understood.
Report Illicit Drug Reaction (RIDR) enables health professionals
to report adverse effects of NPS (or any illicit drug) that they encounter with
patients through a quick and easy to use website https://report-illicit-drug-reaction.phe.gov.uk
and further Information is also available on the RIDR website including summary
reports and publications.
News from the Patient Safety Sub-Group
Pain assessment in people with cognitive impairment
Pain can be difficult to assess in cognitively impaired individuals because their self-report of pain can be inaccurate or difficult to obtain. Hence behavioral and observation–based assessment is required in these patients. Common pain behaviors include: -
- Facial expressions: Frowning, grimacing, distorted expression, rapid blinking
- Verbalizations/vocalizations: Sighing, moaning, calling out, asking for help, verbal abuse
- Body movements: Rigid, tense, guarding, fidgeting, increased pacing/rocking, mobility changes such as inactivity or motor restlessness
- Changes in interpersonal interactions: Aggressive, resisting care, disruptive, withdrawn
- Changes in activity patterns: Appetite change, sleep change, sudden cessation of common routines
- Mental status change: Crying, increased confusion, irritability, distress
Pain can also cause physiological changes such as: -
- Increased heart rate
- Increased blood pressure
- Increased respiratory rate
- Pupil dilatation
Pain assessment tools that combine behavioral and physiological domains for assessment of pain in patients with cognitive impairment: -
- Pain Assessment in Advanced Dementia Scale (PAINAD) - is a pain scale developed by Victoria Warden, Ann C. Hurley, and Ladislav Volicer to provide a universal method of analysing the pain experienced by patients in late-stage dementia.
- Doloplus 2 - is a behavioural pain assessment tool used in elderly patients with limited verbal ability.
- The Abbey Pain Scale - is an is an instrument designed to assist in the assessment of pain in patients who are unable to clearly articulate their needs.
Improving visibility of fentanyl patches
In recent years reports of accidental overdose and unintended transfer of fentanyl patches have been received by the MHRA through their Yellow Card Scheme. Regulatory action was taken in 2016 to improve visibility of Durogesic DTrans patches by insertion of a coloured border around the edge of the patch with different colours to denote the different strengths available. If you are aware of safety incidents relating to accidental overdose of fentanyl please continue to report them to the MHRA; the easiest way to do so is online (www.yellowcard.gov.uk) or through the Yellow Card app.
Safe use of Alfentanil injection
With thanks to the London Opioid Safety & Improvement Group (LOSIG) for providing this information.
Alfentanil is an injectable strong opioid used for pain in patients with severe renal failure (e.g. eGFR <30).
It is administered as single subcutaneous injections, or as a continuous subcutaneous infusion via a syringe pump. It can be prescribed over a wide dose range.
Practice points.
One:
- Two strengths of injection are available: 500 micrograms / mL and 5 mg / mL (10 x stronger).
- Different strengths can be packaged in similar looking boxes.If the wrong box is chosen it can lead to the wrong dose being given.
- When medicines boxes look similar, stop and check the box you pick up is: the right medicine, and the right strength where more than one strength exists.
Two:
- The wide dose range of Alfentanil means that doses can be written in MICROgrams or MILLIgrams (mg).
- If the difference between micrograms and milligrams is not understood it can lead to the wrong calculation results e.g. the volume of the injection to be administered.
- Micrograms are 1000 times smaller than milligrams i.e. 1000 micrograms equals 1 milligram.
- Do not abbreviate micrograms due to the risk of misreading.
Medicines for children
Medicines for Children is a joint project between the Royal College of Paediatrics and Child Health(RCPCH), Neonatal and Paediatric Pharmacist Group (NPPG) and Wellchild which aims to provide reliable practical advice for parents administering medicines to children. The website contains about 200 leaflets and instructional videos. All information is carefully quality controlled and maintained. We encourage you to look at this helpful website and to forward the link below to your interested colleagues, parents and professionals: http://www.medicinesforchildren.org.uk/
 Improving visibility of fentanyl patches
In recent years reports of accidental overdose and unintended transfer of fentanyl patches have been received by the MHRA through their Yellow Card Scheme. Regulatory action was taken in 2016 to improve visibility of Durogesic DTrans patches by insertion of a coloured border around the edge of the patch with different colours to denote the different strengths available. If you are aware of safety incidents relating to accidental overdose of fentanyl please continue to report them to the MHRA; the easiest way to do so is online (www.yellowcard.gov.uk) or through the Yellow Card app.
|
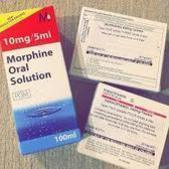 News from the Prescribing Sub-Group
High volume prescribing of morphine sulfate oral
solution
It has come to light
through the work of the prescribing sub-group that some patients are prescribed
very large doses of controlled drugs to manage their pain. In particular we
have seen volumes of morphine sulfate oral solution 10mg /5ml prescribed that
exceeds six litres on a single prescription. While this may be clinically
appropriate for some patients, others may benefit from a review of the
medicines they take and a slow release preparation considered instead. We
highlighted the importance of ongoing review in our recent annual report
and make a recommendation to ensure that the preparation prescribed and the
length of treatment is the most appropriate and effective for the patient to
reduce opportunities for over prescribing and diversion. NHS England CDAOs will
be following this up further in their areas.
This is a sad story that emphasises important lessons when prescribing for pain. Persistent pain is often complex in origin and a full understanding of a patient's physical and emotional health underpins good prescribing. Long term prescribing for pain should always be collaborative between the patient, the GP and specialists involved and clinicians must be mindful of the need for close monitoring and adherence to the treatment plan. Opioid medicines should always be used as part of a broader therapeutic package using non-pharmacological measures to support the patient in improving their quality of life despite ongoing pain.
|
 News from the Vigilance Sub-Group
Good practice points to consider when using the
services of bank & agency staff and advice for lone prescribers
This is an on-going issue that has been raised by members of the
sub-group;
Bank/Agency staff
- Check the relevant register
i.e. GPhC, GMC, NMC and so on for any conditions on their
registration
- Check their PIN number and
identity
- Ask for references
- Where possible, interview them
prior to appointment
- If required, request a DBS
check.
- Do not rely on verbal
assurances from a locum agency.
- Take the time to feedback to
the employment agency if they perform poorly
- Consider whether the locum/bank
member needs to have access to controlled drugs
Beware that those minded to obtain controlled drugs i.e. for
diversion or for their own use can and do apply for jobs in both the health and
care sector.
Be particularly alert to de-registered clinicians, those who have been fired
from roles in healthcare settings and who may have then gone on to change their
name.
Security of prescription forms issued to lone prescribers;
- Completed and blank
prescription forms should be locked in a drawer or cupboard.
- Prescription pads must never be
left in public view.
- Completed and blank
prescription forms carried on vehicles such as ambulances should be locked
in the Trust approved safe. Where vehicles do not have a safe, the
prescription pad should be locked in the boot and the alarm should be
set.
- Where they need to be taken
into a patient’s home, only one or two prescription forms should be taken
in and these must be locked in a case.
Please see NHS Protect's guidance on security of prescription
forms
How to report any unusual behavior that could
be due to the misuse of controlled drugs.
If you have concerns around the theft or misuse of any
controlled drugs either by members of staff or by the public you should report
it straightaway. Timely reporting of concerns can often prevent other crimes or
abuse taking place. There are a number of ways that you can report your concerns:
- Directly report any unusual
activity to the local Controlled Drugs Accountable Officer (CDAO) or NHS
England Lead Controlled Drugs Accountable Officer. CQC's CDAO Register
- Contacting your local Police
Force Controlled Drugs Liaison Officer.
- Report any unusual activity by
calling National Police reporting on 101 or by
- Finding your local police force
website and reporting the unusual activity on-line.
Medicines & Healthcare products Regulatory Agency
(MHRA)’s EU Common Logo
Falsified medicines are a major threat to public health and
safety. Anyone selling medicines to the public via a website must be registered
with MHRA and display an EU common logo on every page of the website offering
medicines for sale. The EU Common Logo (applicable to all legal retailers of
medicines – pharmacies and others) was implemented in UK on 1st July 2015 with
the aim of helping members of the public to identify websites that can legally
sell medicines.
MHRA is the UK authority with responsibility for registering
applicants and compiling a national database with details of suppliers,
premises etc. Further information can be found here and the MHRA press release on selling medicines online
Storage of controlled drugs in a patient's home
Due to the potent nature of controlled drugs, patients, family
members and carers should be given clear instructions on how to store these
medicines securely within their own home. Ideally, advice on safe storage
should be provided by the practitioner issuing the prescription and the
community pharmacist supplying the controlled drug to the patient.
This advice becomes particularly important when young children
are living in the home. A number of children in the UK have died or nearly died
as a result of adults leaving strong analgesics lying around where children can
easily access them. In particular, these medicines can appear very attractive
to a young child who will not know the difference between brightly coloured
medicines and sweets. Methadone along with other controlled drugs, even in very
tiny amounts, can kill a child.
A safety checklist could include some of the following:
- Store controlled drugs &
all medicines safely in the home (locked away, out of sight and
reach)
- Avoid taking medicines in front
of children
- Advice not to put medicines in
bottles without child-resistant lids
- Teach children about the
dangers of medicines
- Never give children medicines
to taste
- Providing information on safe
handling of old or unwanted medicines which includes advice on where to
take for safe disposal
-
Providing advice on storing and where to take
for disposing of injecting equipment safely
|
 Links to key organisations and relevant
Guidance
CQC is the health and social care services regulator with
responsibilities for oversight of safe arrangements for controlled drugs across
England. Website
The Department of Health (DH) helps people to live
better for longer. They lead, shape and fund health and care in England, making
sure people have the support, care and treatment they need, with the
compassion, respect and dignity they deserve. Website
The Home Office leads on immigration and
passports, drugs policy, crime policy and counter-terrorism and works to ensure
visible, responsive and accountable policing in the UK.Website
NHS England leads the National Health Service (NHS) in
England. We set the priorities and direction of the NHS and encourage and
inform the national debate to improve health and care. Website
The National Institute for Health and Care Excellence (NICE)
provides national guidance and advice to improve health and social care. Website
NHS Counter Fraud Authority (NHSCFA)
(formerly known as NHS Protect The NHSCFA)began in shadow form in April
2017. It will be established as an independent special health authority in
autumn 2017. The NHSCFA will provide a clear focus for both the prevention and
investigation of fraud across the health service and will work with NHS England
and NHS Improvement to properly uncover fraud and tackle it effectively. Website
Report NHS Fraud online here or by phoning
the FCRL on 0800 028 4060
Talk to Frank. Offers an educational and confidential advice service on drugs and legal highs. Details can be found here.
|
|