|
Additional CCO Rules RAC added
A new Rules Advisory Committee has been added to discuss CCO rules on September 20, 3 to 5 p.m. Read the proposed rule changes or learn more about HSD Rules Advisory Committees.
Changes to health-related services annual spending report
The OHA health-related services (HRS) team is drafting the 2020 CCO HRS spending report for release in early fall. One change to the annual spending report is that the HRS team will no longer de-identify the spending summary data. What this means is that the report will share total 2020 and 2019 HRS dollar amounts submitted and accepted by OHA as meeting HRS criteria by CCO, as well as per member per month HRS spending and percent of total budget spent on HRS. The report will also identify total dollar amounts spent on HRS flexible services, community benefit initiative, and health information technology by CCO. Please reach out to the HRS team at health.relatedservices@dhsoha.state.or.us with any questions.
As you know, our team has been working on Oregon’s next Medicaid 1115 Demonstration waiver application. Earlier this summer, we released draft concept papers that described our proposed strategies for the waiver application. Since then, we have been revising those drafts in response to robust feedback. After discussion with CMS and an extensive public input process, we have developed a new timeline for both the finalized concept papers and our public comment period. You can see the revised timeline to the right.
The draft concept papers describe the high-level strategies we will pursue in this renewal. So far, public input on the draft concept papers has been robust, and our staff is working to incorporate this feedback. We anticipate making the final concept papers available in multiple languages at the beginning of November and, we will welcome feedback on them.
The waiver application, which is currently being drafted, is a more formal document that outlines which additional federal authorities (permissions) the state is requesting in order to implement the strategies described in the policy concept papers. Prior to submitting the formal waiver application to CMS we will solicit feedback on the application via a 30-day formal public comment period expected to begin in December. During the public comment period, OHA will provide multiple opportunities for the public to provide feedback on the draft application before submitting to CMS.
While this new timeline is slightly delayed from our original ambitious plan, it reflects the reality of incorporating the important feedback we’ve received so far, as well as our commitment to ensuring non-waiver feedback is routed to right programs for consideration.
We will keep you updated as we move forward on this important project. If you have questions, please reach out to 1115Waiver.Renewal@dhsoha.state.or.us.
Thank you,
Jeremy Vandehey, director of Health Policy and Analytics
Lori Coyner, senior Medicaid policy advisor
A memo was posted on 09/13/21 to the CCO Contract Announcements page. Read the full memo.
OHA hereby waives the requirement to publicly post each CCO’s PBM market check report submitted to OHA in Contract Year 2021. This waiver does not relieve the CCO of any other requirement relating to the PBM market check report described in the CCO contract.
-
Exhibit B, Part 2, Section 7, Covered Service Component: Medication Management:Paragraph e.(2)(e)describes the requirement for the CCO to require its Pharmacy Benefit Manager (PBM) to obtain a market check report from an independent third party by July 1stof each Contract Year and for the CCO to submit this report to OHA within 7 days of receipt from its PBM.
-
Exhibit D, Section 14, Transparency: Public Posting of Contractor Reports: Paragraph a. describes the requirement for OHA to publicly post all CCO Reports identified in Exhibit D–Attachment 1as subject to public posting, with approved redactions. The PBM market check report is identified as a Report subject to public posting and redaction.OHA hereby waives the requirement to publicly post each CCO’s PBM market check report submitted to OHA in Contract Year 2021. This waiver does not relieve the CCO of any other requirement relating to the PBM market check report described in the CCO contract.
If you have questions about this communication, please contact Cheryl Henning, CCO Contracts Administrator, at Cheryl.L.Henning@dhsoha.state.or.us.
Please see this excerpt from the FDA's article, "Why You Should Not Use Ivermectin to Treat or Prevent COVID-19."
There seems to be a growing interest in a drug called ivermectin for the prevention or treatment of COVID-19 in humans. Certain animal formulations of ivermectin such as pour-on, injectable, paste, and "drench," are approved in the U.S. to treat or prevent parasites in animals. For humans, ivermectin tablets are approved at very specific doses to treat some parasitic worms, and there are topical (on the skin) formulations for head lice and skin conditions like rosacea.
What you need to know about ivermectin
- The FDA has not authorized or approved ivermectin for use in preventing or treating COVID-19 in humans or animals. Ivermectin is approved for human use to treat infections caused by some parasitic worms and head lice and skin conditions like rosacea.
- Currently available data do not show ivermectin is effective against COVID-19. Clinical trials assessing ivermectin tablets for the prevention or treatment of COVID-19 in people are ongoing.
- Taking large doses of ivermectin is dangerous.
- If your health care provider writes you an ivermectin prescription, fill it through a legitimate source such as a pharmacy, and take it exactly as prescribed.
- Never use medications intended for animals on yourself or other people. Animal ivermectin products are very different from those approved for humans. Use of animal ivermectin for the prevention or treatment of COVID-19 in humans is dangerous.
Other resources about the use of ivermectin
Oregon Health Authority (OHA) is working on more widespread availability of monoclonal antibody (mAb) therapies, which help prevent severe COVID-19 illness, save lives and reduce the burden on our hospitals and health systems.
The federal government announced a new process for distributing mAb products to providers. Health and Human Services (HHS) will now provide allocations through the state, rather than providers ordering directly from the distributor, Amerisource Bergen. OHA is working on a smooth transition to this new process.
Please note, we are currently expecting a limited supply from the federal government and delays in shipments from the distributor.
We know some providers have submitted orders to the distributor that have not been filled. If you are a provider requesting bamlanivimab/etesevimab or casirivimab/imdevimab (RegenCOV) for delivery, please complete this request form by noon on Thursday 9/23. OHA expects to confirm orders by the end of the week.
Federal allocation to the state will be based on utilization, as well as cases and hospitalization rates. Therefore, we need your continued diligence in reporting mAb utilization to ensure Oregon gets the maximum amount of doses available.
Monoclonal antibodies therapy is one more tool to prevent severe COVID-19 disease and support healthy communities, especially while our health systems are under pressure to meet overwhelming need. We encourage all providers to offer mAb therapy, particularly now that it is approved for both subcutaneous and intravenous application. We appreciate your patience through this transition.
For more information, please visit OHA’s mAbs webpage. Questions? Contact ORESF8.LogisticsChiefs@dhsoha.state.or.us.
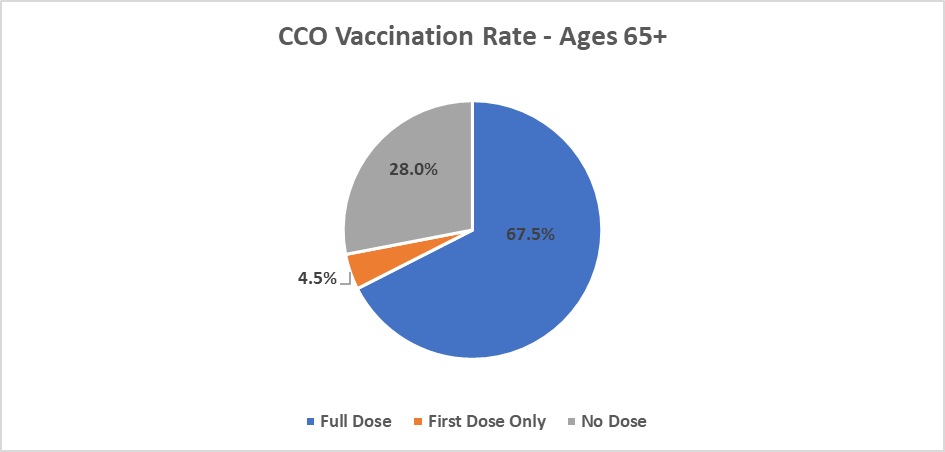
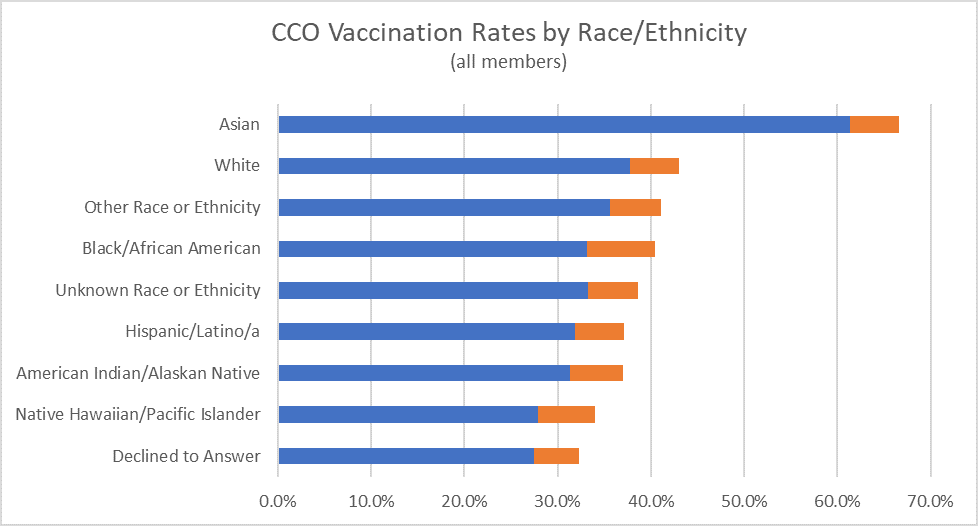
CCO COVID Vaccination Rates as of 9/15/2021
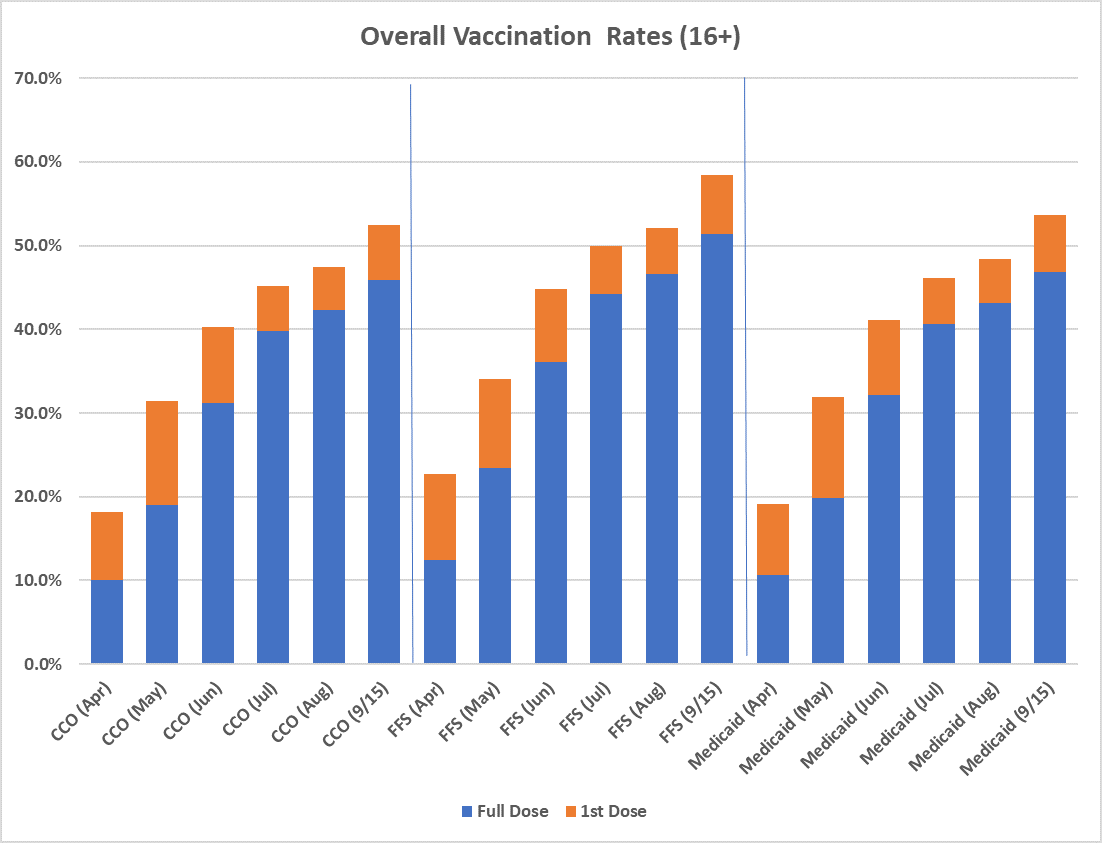 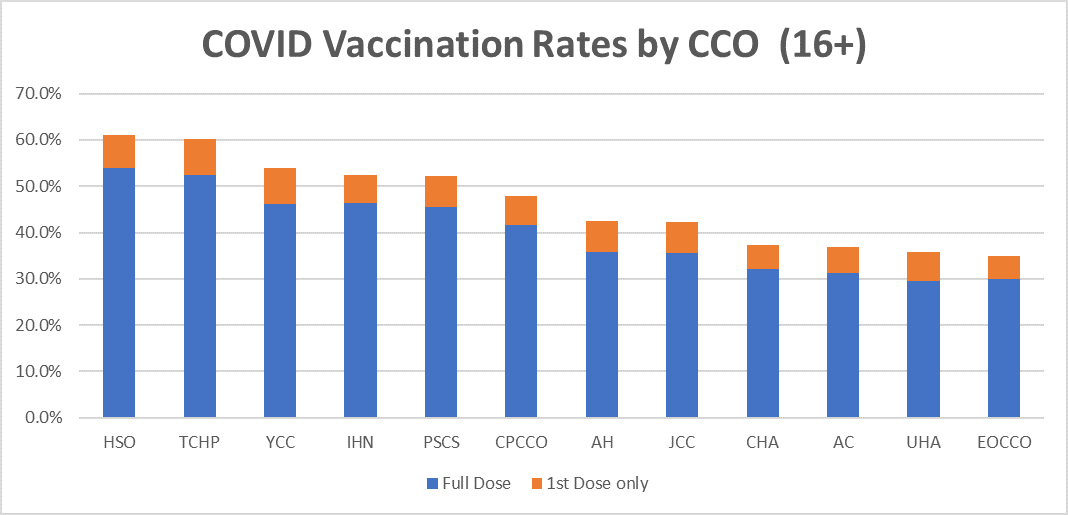 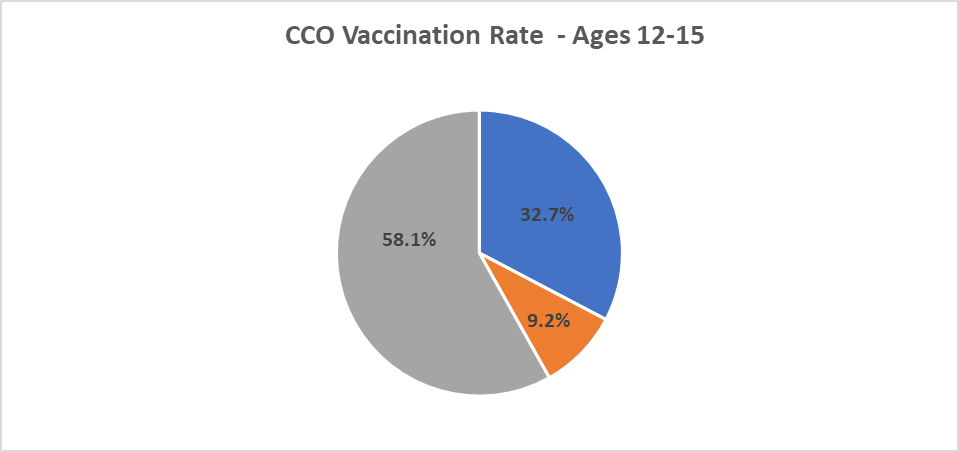 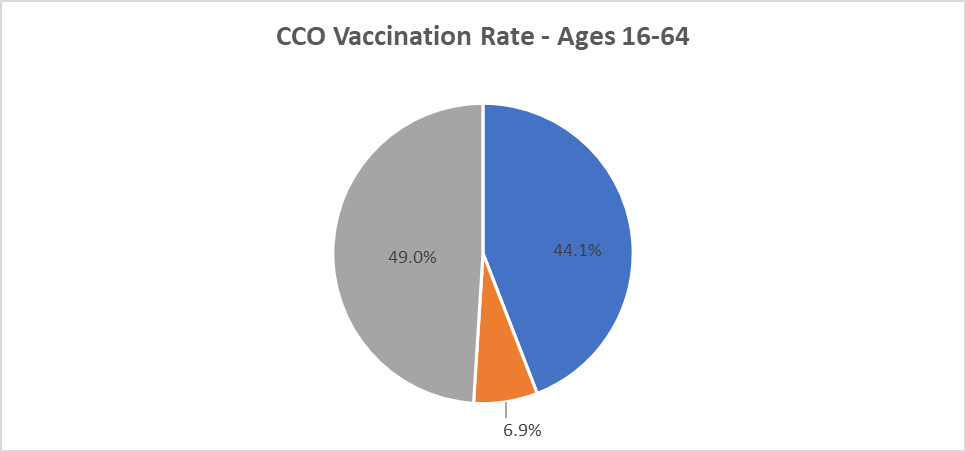  
Please visit the committee and workgroup links below for more information about meetings. You can also view the OHA Public Meeting calendar.
|