|
There are several reasons besides inappropriate age or interval that a dose may be “NOT VALID” on a client’s MIIC record. Doses administered from vaccine lots associated with storage and handling issues, recalled vaccine, or expired lots can lead to sub-optimal immune responses and require the person to be revaccinated. As with other “NOT VALID” doses in a person’s immunization history, the MIIC forecaster will not count these doses towards the recommended vaccination schedule which may also impact doses received after the not valid vaccine. The picture below depicts an invalid dose of COVID-19 vaccine in MIIC and the recommended schedule for a client. Please contact the MIIC helpdesk with questions: health.miichelp@state.mn.us.
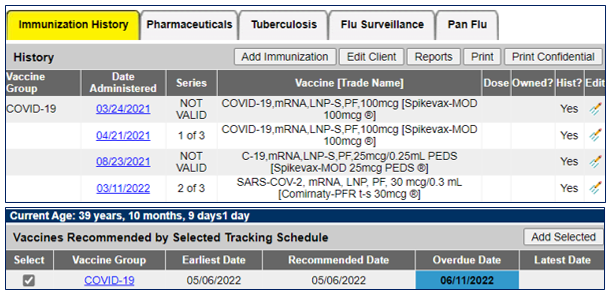
Providers that encounter a situation where a patient’s MIIC record indicates that a COVID-19 dose is “NOT VALID” should assess whether the person has completed their COVID-19 primary series. If a person does not have a completed primary series or if their primary series was invalidated due to a “NOT VALID” dose, the client usually needs an additional monovalent vaccine dose for their vaccine record to show a valid primary series. Having a valid primary series that uses the original vaccine products produces foundational immunity that cannot be gained from the bivalent product and provides a broader range of immune response.
Before administering any COVID-19 vaccine, it is important to assess every client’s immunization history in MIIC.
Does the client have a valid primary series?
- No: Finish their primary series by administering a monovalent COVID-19 vaccine.
- Yes: Has it been at least two months since the most recent dose?
- Yes: Administer a bivalent or age-appropriate* booster if they haven't received it.
- No: Wait to give the bivalent until at least 2 months after primary series completion.
*People 5-11 years of age are indicated to receive a monovalent booster at this time.
If you have questions regarding a person’s COVID-19 immunization history or revaccination, please email health.covid.vaccine@state.mn.us.
There are a lot of reasons to get a COVID-19 vaccine, and we want to hear from Minnesotans what some of those reasons are.
We’re asking people to share why they got themselves or their children vaccinated for COVID-19 to help encourage others to do the same. Maybe you recently got your updated bivalent booster to be better protected before seeing vulnerable family members for holidays in a couple of months. Maybe you wanted to make sure your children were protected for a new school year. Whatever your reason for getting vaccinated, we want to hear from you.
You can record a short video to submit to MDH and we will help share some of the reasons people are rolling up their sleeves.
Visit Share Your Story: Why I Got the COVID-19 Vaccine for more information and instructions for submitting a story.
|