|
View this as a webpage
Minnesota Department of Health
October 28, 2021
Good afternoon,
Here are the COVID-19 vaccine provider updates for Oct. 22 to Oct. 28, 2021.
CDC’s Interim Clinical Considerations for Use of COVID-19 Vaccines was updated on Oct. 25, 2021, to reflect new recommendations for booster dose administration of the Pfizer-BioNTech, Moderna, and Janssen (Johnson & Johnson) COVID-19 vaccines. Recommendations for use of a single COVID-19 booster dose after completion of a primary series can be found in the Overview of COVID-19 vaccine recommendations.
Here is a summary of the most recent changes:
Following the FDA’s expansion of the EUA for booster doses, Moderna sent a letter to alert vaccination providers that the volume of the booster dose is 0.25 mL; half the dose administered in the primary series. We highly encourage you to read the document in its entirety: Moderna HCP letter: Important Prescribing Information (PDF).
If you missed it, please refer to our earlier communication sent on Oct. 22, 2021, for a detailed summary of the interim recommendations for COVID-19 vaccine booster doses at COVID-19 Vaccine Provider Updates.
People with Down Syndrome are at an extremely high risk for severe COVID-19 disease. Our Disabilities Team at the health department wants to make sure our partners are aware of all the data coming out about this. This is especially pertinent to vaccine providers as we roll out booster doses to those at risk.
The provider guide and appendices have been updated with current information on COVID-19 vaccine booster doses. Find them at COVID-19 Vaccine Providers.
- Provider guide: added MIIC texting project to second dose section (p. 16), updated booster dose section (p. 17), updates to vaccine efficacy section (p. 18).
- Appendix A: updated booster dose recommendations (p. 6).
- Appendix B: updated shipping content (pp. 1-2), updated booster dose recommendations (pp. 4-5), updates to vaccine preparation (p. 9).
- Appendix C: updated thawing information (p. 1), updated booster dose recommendations (p. 3).
All three vaccine protocol templates are updated on the Vaccine Protocols webpage and at COVID-19 Vaccine Providers.
By the end of the week, MIIC will forecast a booster dose for clients age 65 and older who have received two doses of an mRNA COVID-19 vaccine (Pfizer-Comirnaty or Moderna-Spikevax). The recommended date will be six months after the date of the second dose. Since CDC has removed the requirement for product specificity, all three currently approved U.S. vaccines will be valid. In addition, MIIC will now forecast a booster dose for all clients 18 years and older who have received a dose of the Janssen COVID-19 vaccine. The recommended date will be two months (56 days) after the date of the initial dose. If you have any questions regarding the forecaster in MIIC, please contact the MIIC Help Desk.
Join Moderna for a webinar for vaccination providers to learn more about the Moderna COVID-19 vaccine booster dose. There will be no continuing education offered for this webinar.
Please register at the link below for one of the available sessions:
When extracting only booster doses, or a combination of primary series and booster doses, from a vial, the maximum number of doses that may be extracted from either vial presentation should not exceed 20 doses. Do not puncture the vial stopper more than 20 times. Wastage should be reported only as whole doses. Each dose administered, whether it is a half dose or a full dose, counts against the total possible wastage of 10 doses (for Moderna 10) or 14 doses (for Moderna 14).
- For a Moderna-14 vial, wastage should only be reported up to 14 doses; do not report wastage over 14 doses, even though you can administer up to 20 booster doses from one vial.
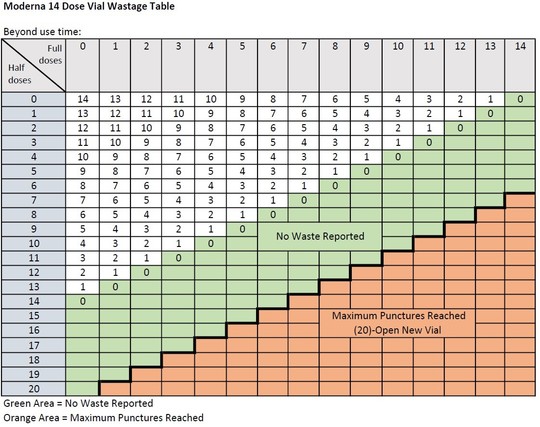
- For a Moderna-10 vial, wastage should only be reported up to 10 doses; do not report wastage over 10 doses, even though you can administer up to 20 booster doses from one vial.
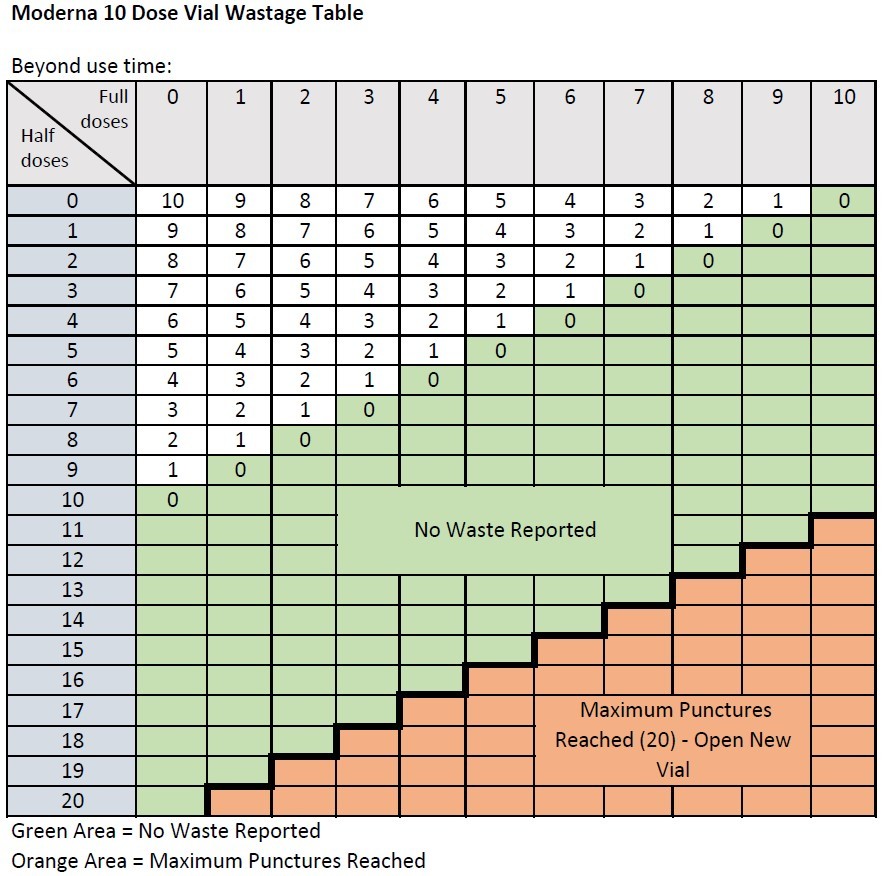 This information will also be updated in our user guide Reporting Nonviable COVID-19 Vaccine to MIIC (PDF) soon.
On October 26, FDA's Vaccines and Related Biological Products Advisory Committee (VRBPAC) voted to recommend FDA authorization to expand use of the Pfizer-BioNTech COVID-19 vaccine for children ages 5-11 years. Specific authorization from FDA is pending and the CDC’s Advisory Committee on Immunization Practices (ACIP) meets next week to discuss more specific guidance. Do not begin vaccinating until ACIP makes its recommendations and CDC issues their formal guidance.
Details on the vaccine
The vaccine for this age group is packaged differently than the current adult/adolescent vaccine (for people down to age 12 years) because younger-aged children will be receiving a smaller dose. It will not be acceptable to make a pediatric dose from the adult vaccine.
The 5–11-year-old pediatric vial will have an orange label and an orange cap and will have different storage and handling requirements than the adult/adolescent vaccine currently in the field. The product can be kept in the ultra-low cold freezer for up to six months and in the refrigerator for up to 10 weeks. This product CANNOT be stored in the regular freezer. The pediatric vaccine will come in 100 dose packages (10 multi-dose vials without preservative with 10 doses per vial). This vaccine is shipped in smaller thermal shippers, which cannot be used for vaccine storage.
Timing of next MIIC pediatric vaccine ordering events
The pre-book event for the first week of the pediatric COVID-19 vaccination campaign is now closed. The next ordering event for pediatric COVID-19 vaccines will open on Friday, November 5, and will close at 4:30 p.m. on Monday, November 8. Orders will be processed by Wednesday, November 10. Thereafter, ordering events for pediatric COVID-19 vaccine will open on Mondays and close at the end of the day (4:30 p.m.) on Tuesdays. Minimum order sizes will be 100 doses. You must be enrolled in the COVID-19 vaccine program to order pediatric COVID-19 vaccine. Pharmacies enrolled in the Federal Retail Pharmacy Program (FRPP) should request doses via their CDC FRPP contacts.
Shipping
We anticipate shipping for the initial doses will begin once the emergency use authorization (EUA) is granted and will continue over the course of that week. Please check your email for shipping confirmation emails from Pfizer. Because of the rolling nature of orders, some sites may receive shipments a couple days before others. Reminder: You may not begin administering the pediatric COVID-19 vaccine until ACIP makes a recommendation and CDC issues formal guidance. ACIP is expected to meet the first week of November to discuss the pediatric COVID-19 vaccine.
Please note that second doses will not automatically be shipped. You must manage your inventory for second doses and request first and second dose needs in future weeks. Pfizer expects shipping timelines to remain the same (three-five days for shipment once order is placed).
Pfizer adult/adolescent vaccine ordering temporarily suspended
In order to maximize delivery of pediatric doses, adult/adolescent Pfizer COVID-19 vaccine ordering was suspended as of 8 a.m. this morning and will be suspended through Wednesday November 3, 2021. No adult/adolescent Pfizer vaccine will be shipped the week of November 1-5.
Pfizer U.S. Medical Affairs is hosting “Medical Updates” for its pediatric COVID-19 vaccine. Topics include FDA indication & authorization, CDC/ACIP recommendations, packaging/presentation, storage and handling, and a Q&A session.
Each session will be unique and will reflect any new information or changes that emerge. Click on the links below to join the sessions at the designated times.
Health care providers and partners, join Hennepin HealthCare on Monday, November 1 at noon for a webinar titled: Helping Kids Cope with Vaccinations; the Children's "Comfort Promise.” Donna Eull, MA, PhD, RN, CHPPN, with Children's Minnesota, will help participants look at current practices with some of the advancements in vaccine delivery to children and engagement of their parents and guardians. This presentation will provide practical tips and resources including preparation, language, engaging parents, and use of evidence-based strategies to improve the patient experience.
To join the webinar: https://HennepinHealthCare.zoom.us/j/83632589909
Or one tap mobile: US: 8884754499,,83632589909# (Toll Free) or 8778535257,,83632589909# (Toll Free)
Or telephone: Dial (for higher quality, dial a number based on your current location): US: 888 475 4499 (Toll Free) or 877 853 5257 (Toll Free) Webinar ID: 836 3258 9909 International numbers available: Zoom International Dial-in Numbers
Keeping parents up to speed on when and why their child should get vaccinated is critically important to keeping children safe and slowing the spread of COVID-19. Please help us share information about the safety of the vaccine and why it is important for children to get vaccinated with families in your social circles.
We have several resources on our website that may help you (Translations available at About COVID-19 Vaccine):
There are two online tools designed to help people find vaccine in their community. With the expected authorization of COVID-19 pediatric vaccine next week, we encourage providers to register and publicly display their locations on both the federal VaccineFinder website and the state’s Find Vaccine Locations map. This will help parents and caregivers easily find vaccine for their 5-11-year-olds.
VaccineFinder (Vaccines.gov) is an optional federal tool that can direct people to COVID-19 vaccine providers that have vaccine available. You were invited to sign up when you registered to be a COVID-19 vaccine provider; this is a friendly reminder to make sure your site is “public.” MDH will continue to report inventory on behalf of all sites (except Federal Retail Pharmacy Program participants). Once your site is registered with VaccineFinder, please refer to the Quick Start Guide: VaccineFinder Public Display Administration Fields – Log Manually (PDF) for instructions on how to update your public display information. For any questions, or to be resent an invitation to participate, you can contact their help desk: vaccinefinder@castlighthealth.com or visit CDC’s page Vaccines.gov Public Display Checklist for Providers using the COVID Locating Health Provider Portal.
If you have not yet done so, please email health.covid.vaccine@state.mn.us to publicly list your site on the state’s Find Vaccine Locations map.
Remember to include vaccine lot number in addition to client name, date of birth, date of administration, and vaccine product type on the CDC COVID-19 vaccination card. It is important for the card to be filled out completely, as some people are asked to present the card as verification of having received COVID-19 vaccine.
Seventy-eight lots of Moderna vaccine received a shelf-life extension from FDA. However, Moderna has been very slow at entering the updated expiration dates into their site. Most of the vaccine that expired in September and October may have an additional two-month extension. Please continue to check the Moderna expiration date lookup site at Moderna: How To Look Up Vial Expiration Date before disposing of presumably expired Moderna vaccine to see if your product is included in this set of lots. You can also confirm all expiration dates using the QR code on the Moderna vial. The extensions will only apply to vaccine still in the freezer. If the vaccine has already been moved to the fridge, it expires on the original expiration date.
Please refer to Moderna’s website for more information. If the website does not reflect these changes yet, Moderna has said it should be up to date soon. Consider ordering new Moderna vaccine if your vaccine is expired, and you are waiting for them to add in your lot.
If you are still awaiting an expiration extension of your lot, mark any expired Moderna vials “DO NOT USE.”
Expired/expiring Moderna doses
The listed batches of Moderna vaccine in this PDF are not subject to an extension of their expiration date: ModernaVaccineNOTSubjecttoExpirationExtension.pdf. Prioritize unexpired lots (listed below) and use according to their expiration dates. Please try to use all doses prior to expiration to help reduce wastage.
Expired lots on this list must not be used and should be discarded.
|