|
View this as a webpage
Minnesota Department of Health
August 26, 2021
Good afternoon,
Here are the COVID-19 vaccine provider updates for Aug. 20 to Aug. 26, 2021.
Public health and medical experts from the U.S. Department of Health and Human Services (HHS) released information on initial plans for COVID-19 vaccine booster doses among adults on Aug. 18, 2021. Booster doses are currently not recommended. Both U.S. Food and Drug Administration (FDA) authorization and updated recommendations from the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices (ACIP) need to occur before that happens. Read the HHS press release at Joint Statement from HHS Public Health and Medical Experts on COVID-19 Booster Shots.
Giving booster doses without FDA authorization and ACIP recommendation are a violation of your provider agreement and may put you at risk of no longer being able to provide vaccine.
Continue your current focus to get unvaccinated patients vaccinated and complete vaccination series in partially vaccinated people. This focus has the greatest impact on reducing the spread of COVID-19 and its more severe outcomes. Nearly all the cases of severe disease, hospitalization, and death continue to occur among those not yet vaccinated. The COVID-19 vaccines authorized in the United States continue to be remarkably effective in reducing risk of severe disease, hospitalization, and death, even against the Delta variant.
As a reminder to providers, a booster dose is different from the recent ACIP recommendation to give an additional dose of mRNA COVID-19 vaccine after an initial primary vaccine series for certain immunocomprised people. Additional information is published in the CDC Interim Clinical Considerations for Use of COVID-19 Vaccines: Considerations for use of an additional mRNA COVID-19 vaccine dose after an initial 2-dose COVID-19 mRNA vaccine series for immunocompromised people.
FDA approved the Pfizer-BioNTech COVID-19 vaccine on Aug. 23, 2021 for prevention of COVID-19 disease in people ages 16 and older. This mRNA vaccine will now be marketed by the brand name, Comirnaty. The vaccine continues to be available for people ages 12-15 under emergency use authorization (EUA). Read more in the news release at FDA Approves First COVID-19 Vaccine.
COVID-19 vaccine providers should continue to vaccinate and use Pfizer COVID-19 vaccine as you have been. The full license does not impact current recommendations. ACIP will be meeting on Monday, Aug. 30, 2021, to discuss updating its recommendation for this vaccine.
While we wait for a vaccine to be authorized in children younger than age 12, it is important that everyone who is eligible now get the vaccine. The Delta variant has led to significant increases in the number of children and adults infected with COVID-19. Vaccination of eligible groups will help reduce the spread of the virus and protect those who are too young to be vaccinated.
Off-label actions
Off-label actions (such as administering the vaccine to children ages 11 and younger) are a violation of section 1 of the COVID-19 Vaccination Program Provider Agreement. As a reminder, providers are responsible for adhering to all requirements outlined in the agreement. Specifically, providers must administer COVID-19 vaccines in accordance with all CDC COVID-19 Vaccination Program Provider Requirements and Support of CDC, ACIP, and FDA. This applies to both EUA and FDA-approved COVID-19 vaccines. Uses of these products outside of those that have been approved and authorized by FDA (off-label use”) could expose providers to the following risks:
- Administration of the product off label may not be covered under the PREP Act or the PREP Act declaration; therefore, providers may not have immunity from claims.
- Individuals who receive an off-label dose may not be eligible for compensation under the Countermeasures Injury Compensation Program after a possible adverse event.
- CDC has defined the scope of the CDC COVID-19 Vaccination Program in terms of how the USG-provided vaccines may be used in the program. Providers giving off-label doses would be in violation of the CDC program provider agreement potentially impacting their ability to remain a provider in the CDC program.
- Administration fees may not be reimbursable by payers.
The American Academic of Pediatrics strongly discourages providers from administering the vaccine off-label for children ages 11 and younger. Clinical trials are underway for this age group and more data will hopefully be available soon. Read more at American Academy of Pediatrics Cautions Against Off-Label Use of COVID-19 Vaccines in Children Under 12.
Updated EUA fact sheets
FDA Provider and Recipient/Caregiver Fact Sheets have been updated following the FDA’s approval of a Biologics License Application (BLA) for Pfizer’s Comirnaty COVID-19 Vaccine. The BLA approval is for adults 16 years and older; the EUA for adolescents will continue to cover adolescents ages 12-15. At this time, it has been decided to continue using the EUA fact sheet format in the near term instead of a vaccine information statement (VIS) document. The FDA amendments and BLA documents for Pfizer have been updated as of Aug. 23, 2021. Find them at Comirnaty and Pfizer-BioNTech COVID-19 Vaccine.
The FDA has approved an amendment to the EUA for Pfizer-BioNTech COVID-19 vaccine, extending the expiration dates from six to nine months.
Cartons and vials of Pfizer-BioNTech COVID-19 vaccine with an expiration date of August 2021 through February 2022 printed on the label may remain in use for three months beyond the printed date, as long as authorized storage conditions between -90 to -60 degrees Celsius (-130 to -76 degrees Fahrenheit) have been maintained.
Please note: the ultra-cold temperature range has been broadened to include -90 degrees Celsius (-130 degrees Fahrenheit). Frozen vials stored at -25 to -15 degrees Celsius and refrigerated vials (2 to 8 degrees Celsius) are not eligible for extension.
Updated expiry dates for vaccine maintained in ultra-cold storage are shown below.
|
Printed expiry date
|
Updated expiry date
|
|
August 2021
|
November 2021
|
|
September 2021
|
December 2021
|
|
October 2021
|
January 2022
|
|
November 2021
|
February 2022
|
|
December 2021
|
March 2022
|
|
January 2022
|
April 2022
|
|
February 2022
|
May 2022
|
The extended expiration date is effective immediately for all currently available batches that have not yet expired.
Beyond-use date (BUD): Keep in mind that vaccine with extended expiration dating that has been stored at ultra-cold or freezer temperatures and recently moved to the refrigerator can be used through the BUD date, even though the original expiration date may be shorter. An example would be, vaccine that was moved to the fridge on Aug. 15, 2021 and had an original expiration date of Aug. 31, 2021 now has an expiration date of Nov. 30, 2021, so can be used through Sept. 15, 2021, instead of discarding it on Aug. 31, 2021 when it would have originally expired.
Note: Expiration date extension does not apply to vials dated July 2021 and earlier.
Additional information on Pfizer storage and handling may be found at: CDC: Pfizer-BioNTech COVID-19 Vaccine.
Pfizer will again be hosting training sessions to review information and answer questions about the Pfizer-BioNTech COVID-19 vaccine.
Please click on the links below to join the sessions at the designated times. Each training below is the same content.
COVID-19 vaccine protocol updates
Pfizer-BioNTech and Moderna COVID-19 vaccine protocols were updated to include the additional dose for immunocompromised people and can be found on the Vaccine Protocols webpage and at COVID-19 Vaccine Providers under "Forms and templates."
MDH COVID-19 Screening and Agreement template updated
The screening and agreement template has been updated at COVID-19 Vaccine Providers under "forms and templates."
COVID-19 vaccine provider guide and appendices
Find updated documents at COVID-19 Vaccine Providers.
- Main provider guide: updates for FDA approval of Pfizer COVID-19 vaccine (p. 4), updated training requirements (p. 5), other minor updates throughout.
- Appendix A (Pfizer-BioNTech): shipping updates (p. 3), storage updates (pp. 3-4), updated recommendations (p. 5).
- Appendix B (Moderna): updated recommendations (p. 3).
Due to the federal holiday, there will be no Pfizer deliveries on Monday, Sept. 6, and there will be no Moderna deliveries on Monday, Sept. 6 and Tuesday, Sept. 7.
Ordering in MIIC
Providers can directly request COVID-19 vaccines in the Minnesota Immunization Information Connection (MIIC) at any time. We will process orders daily. Sites can expect to receive doses within two-three business days after submitting their order. Please contact health.mdhvaccine@state.mn.us with questions.
Refer to our user guide for step-by-step instructions on how to order COVID-19 vaccine in MIIC: User Guide: Special Event Vaccine Request (PDF).
Over the next several weeks, a changing variety of existing COVID-19 vaccine product presentations (i.e., different packaging options) will be available to request in MIIC. CDC is making an effort to distribute all remaining COVID-19 product presentations while we await the next federal contracts to renew later this fall.
Prioritize administering vaccine doses you have on hand and avoid stockpiling doses. Order only what you can use for the next couple of weeks.
Supply updates
Supply of Moderna and Pfizer COVID-19 vaccines remains plentiful.
Pfizer vaccine
- The 450 packs of Pfizer are no longer available. The national supply of this popular package size has run its course.
- 1,170 packs of Pfizer vaccine remain available in MIIC.
- Ancillary kits for the 1170 packaging are configured with 80% 1-inch needles that may be used for pediatric patients.
- If you would like to request Pfizer vaccine in an amount less than 1,170 doses, please visit our Smartsheets tool: COVID-19 Vaccine Available for Redistribution.
Moderna vaccine
- Moderna 14-dose vials remain available.
- Moderna 10-dose vials are back. CDC expects inventory of the 10-dose vials to phase out in the next few weeks. All 10-dose vials expire in November/December 2021. Please visit our Smartsheets tool: COVID-19 Vaccine Available for Redistributionbefore submitting requests in MIIC; currently there are more than 20,000 doses of Moderna 10-dose vials ready for redistribution in the state.
Janssen (Johnson & Johnson) vaccine
- Currently not available for requesting in MIIC. We will let you know when this is available for ordering. CDC anticipates Janssen vaccine becoming available in early September. All upcoming supply of Janssen vaccine will have a six-month shelf life.
On Aug. 13, ACIP recommended a third dose of mRNA-containing COVID-19 vaccine for moderately to severely immunocompromised people who have completed the two-dose mRNA COVID-19 vaccine series with either Pfizer (Comirnaty) or Moderna (Spikevax). Find additional information in the CDC Interim Clinical Considerations for Use of COVID-19 Vaccines: Considerations for use of an additional mRNA COVID-19 vaccine dose after an initial 2-dose COVID-19 mRNA vaccine series for immunocompromised people.
Because the third dose is recommended for a small subset of the population, MIIC will not predict a third dose of vaccine. Once administered, the dose will appear in the client’s MIIC immunization record. Below is an example of how it will appear in MIIC.
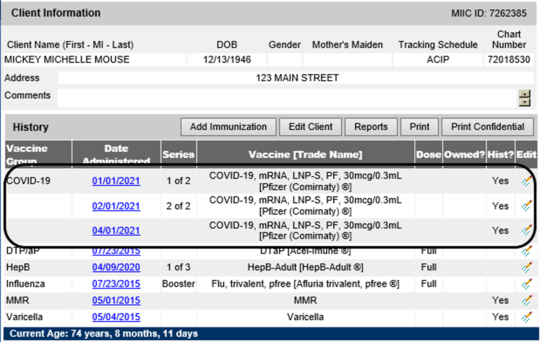 The MIIC data quality team will continue to work with organizations to review their data for clients with three documented doses of COVID-19 vaccine in MIIC and determine if any of these doses were entered incorrectly. It is increasingly important to have completed this cleanup, given new recommendations around third doses and possible recommendations around booster doses. If you have any questions, please reach out to the MIIC help desk.
It continues to be best practice to have an observation period following vaccination for any vaccine being given. This is especially important with the COVID-19 vaccines, due to the increased incidence of allergic reactions. This does not change even if you are giving a third dose, as the potential for severe allergic reactions continues, regardless of how many doses people have received.
As more schools, businesses, and venues require documentation of COVID-19 vaccination, it is important to remember that data in MIIC contain private information about people that must be treated in a manner that preserves their privacy. Organizations must complete and submit a MIIC Data Use Agreement (DUA) before being set up in MIIC, and every three years while participating, as a commitment to upholding data privacy and security. As part of the DUA, organizations agree to “access, provide and share immunization data only as allowed by the Minnesota Immunization Data Sharing Law (Minn. Stat. §144.3351).” Per state law, organizations can only view a person’s MIIC record, without their consent, if the organization provides services on behalf of that person. Discuss with your organization’s administrative and/or legal teams to determine how this law applies to your organization’s use of MIIC. The Minnesota Department of Health (MDH) cannot provide legal guidance. Unauthorized disclosures by private providers are governed by Minn. Stat. §144.298, which allows for “disciplinary action against a provider by the appropriate licensing board or agency.”
MDH does not have CDC vaccination cards. Please do not direct patients to call MDH for these cards. Your organization can provide your patient with a copy of their vaccination or MIIC record. Learn more in the Client Search and Printing Immunization Records (PDF) user guidance. Patients who contact MDH requesting record of their vaccination are provided a printout of their MIIC record. Record requests are typically filled within 10-14 business days.
For those who could not join the webinar on adolescent catch-up immunization with the MDH immunization team, Dr. Chomilo, and Dr. Singh, you can now review the content presented: Adolescent COVID-19 Vaccine Co-Administration Webinar Notes.pdf.
|