 CDC Shortens Isolation and Quarantine Recommendations
On December 27, 2021, the Centers for Disease Control and Prevention (CDC) issued updated guidance which shortened the recommended isolation period for someone with COVID-19, as well as quarantine guidance for those exposed to someone with COVID-19. You can read the CDC’s statement on the changes here. The Ottawa County Department of Public Health will modify its isolation and quarantine requirements and recommendations, effective immediately. We expect our communications, policies, procedures, and systems to be updated with this guidance within the next week. In addition, we are in the process of reviewing and updating our school guidance and orders to align more closely with the updated CDC recommendations. Visit miottawa.org/COVID19 for more information.
READ MORE

FDA Authorizes First Oral Antiviral for Treatment of COVID-19
On December 22, 2021, The Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for Pfizer's Paxlovid™ pill. The oral antiviral pill is intended for the treatment of mild-to-moderate COVID-19 in adults and pediatric patients over age 12 who have risk factors for severe COVID-19, hospitalization or death.
“This authorization provides a new tool to combat COVID-19 at a crucial time in the pandemic as new variants emerge and promises to make antiviral treatment more accessible to patients who are at high risk for progression to severe COVID-19,” Dr. Patrizia Cavazzoni, director of the FDA’s Center for Drug Evaluation and Research, said in a statement on Paxlovid™, marking the introduction of the first COVID-19 oral treatment pill.
Paxlovid™ is available by prescription only and should be initiated as soon as possible after diagnosis of COVID-19 and within five days of symptom onset. Paxlovid™ is administered as three tablets taken together orally twice daily for five days, for a total of 30 tablets. There are side effects associated with the drug's use and it is not recommended for all patients. If you test positive for COVID-19, contact your health care provider immediately to see if you are eligible for treatment with Paxlovid™.
READ MORE
On December 23, 2021, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for Merck’s molnupiravir for the treatment of mild-to-moderate coronavirus disease (COVID-19) in adults with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death, and for whom alternative COVID-19 treatment options authorized by the FDA are not accessible or clinically appropriate. Molnupiravir is available by prescription only and should be initiated as soon as possible after diagnosis of COVID-19 and within five days of symptom onset.
Molnupiravir is not authorized for use in patients younger than 18 years of age because molnupiravir may affect bone and cartilage growth. It is not authorized for the pre-exposure or post-exposure prevention of COVID-19, or for initiation of treatment in patients hospitalized due to COVID-19 because benefit of treatment has not been observed in people when treatment started after hospitalization due to COVID-19.
READ MORE
Why We Need to Upgrade Our Masks Now That Omicron is Here
With cases of the highly contagious Omicron variant on the rise, experts are recommending upgrading cloth masks to something more protective, ideally an N95 or similar mask.
At the beginning of the pandemic, health officials did not recommend KN95 or N95 masks due to the shortage of supply of these types of masks for health care workers. Now, KN95 and N95 masks are widely available. In addition, we have learned more about how the coronavirus spreads through droplets in air shared between people. Cloth masks may not be effective at preventing the larger load of particles from entering your nose and mouth. N95 and KN95 masks fit more snugly, are durable, and are better at preventing air (which may contain virus particles) from leaking out around the edges. Health experts are now recommending well-fitted, high filtration masks, especially in indoor public settings.
If you do not have access to a high filtration mask, consider a 3-ply surgical mask, or consider pairing a disposable mask with a cloth mask.
The National Personal Protective Technology Laboratory (NPPTL) provides tips on how to spot a counterfeit N95 masks and Project N95 offers vetted sources for personal protective equipment.
Read more:
CDC Your Guide to Masks
Scientific American
Early Treatment Options Available for COVID-19
We are often asked about the availability of early treatments for COVID-19, when an individual can receive therapies as soon as possible after being diagnosed with COVID-19, in order to potentially shorten the duration and severity of symptoms of the disease. Currently, there are only two forms of early treatment authorized for use in treating COVID-19 outside of a hospital setting.
- Monoclonal Antibody Treatments - Monoclonal antibodies are laboratory-produced molecules that act like the antibodies the immune system produces to fight a particular infection. Monoclonal antibodies for COVID-19 may block the virus that causes COVID-19 from attaching to human cells, making it more difficult for the virus to reproduce and cause harm.
- Antiviral pills under emergency use authorization: Paxlovid (Pfizer) and Molnupiravir (Merck) (see descriptions above)
READ MORE
While overall good health, and things like vitamins, supplements and a healthy diet can influence how an individual experiences COVID-19 infection, there is no evidence that these prevent or treat COVID-19 infection, or any infectious disease.
"Health care providers and public health departments have consistently promoted healthy behaviors, including good nutrition, and haven't stopped these messages during the pandemic," said Marcia Mansaray, Deputy Health Officer at Ottawa County Department of Public Health. "While any number of things have been demonstrated to contribute to general good health, like vitamin D3, at this time there is no body of evidence that any of those things prevent COVID-19 infection or treat COVID-19 infection. If there are well-designed, peer-reviewed studies that establish a sound body of evidence that treatments prevent or treat an infectious disease, our public health and healthcare providers will promote them."
One way scientists and health care providers learn what treatments are safe and effective is through clinical trials. If you are interested in participating in a trial that may help scientists and healthcare providers learn more about how to safely and effectively treat COVID-19, check out the information here or here.
Questions & Answers
What is health misinformation?
Public Health has been defined as the science of protecting and improving the health of people and their communities. A challenge many public health officials, doctors and health care providers, scientists and others doing the work of protecting community health have faced throughout the pandemic is the spread of misinformation and disinformation. The false information is sometimes spread purposefully, and is often broadly shared on social media platforms, often by those who do not know they are sharing inaccurate information.
U.S. Surgeon General, Dr. Vivek H. Murthy, has prioritized addressing health misinformation because it "harms people's health, and undermines public health efforts". This summer, Dr. Murthy released an advisory on the urgent threat of health misinformation and a toolkit about how we can all be better educated individually, and help shape our information environment to build a "healthier, kinder and more connected world".
Your health, and the health of your friends and family, matters. OCDPH wants to help equip you to find scientific, understandable and credible information you can use to inform your health decisions. Use the Community Toolkit for Addressing Health Misinformation to understand what health misinformation is and how to identify it.

Kids-Only COVID-19 Vaccine Clinics Available in January
We've added more kids-only vaccine clinics in January. If you're still looking for a vaccine for your child, scan the QR code at the clinic of your choice below to register for an appointment. As a reminder, Ottawa County's school mask mandate will expire on January 2, 2022. Vaccination continues to be the best way to protect your children from serious COVID-19 illness as they head back to the classroom. We know parents might have questions about whether the vaccine is safe and effective for children. The American Academy of Pediatrics discussed vaccine safety here. In addition, we encourage you to speak to your pediatrician or physician for more information.
 

Ottawa County Data Hub
Ottawa County publishes recent COVID-19 data relating to community transmission, counts, descriptive statistics and test positivity. The data hub is updated every weekday from Monday - Friday by 4:00 pm, but will not be updated this Friday, December 31 in observance of the New Year's Holiday. View Data Hub
Weekly COVID-19 Data Reports can be found on our COVID page.
Ottawa County Case Rates
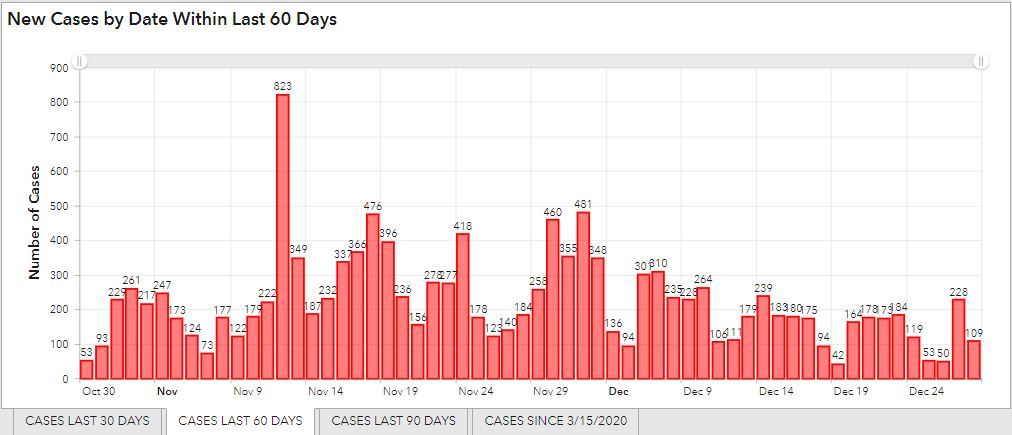 As of December 29, 131 new cases of COVID-19 were reported each day on average over the last 7 days, similar to the 132 daily reported cases over the previous 7-day period. Case counts and incidence rates remain high at this time. Because these reports do not include results in people tested with antigen test kits at home, true case counts may be underreported.
COVID-19 Vaccinations in Ottawa County
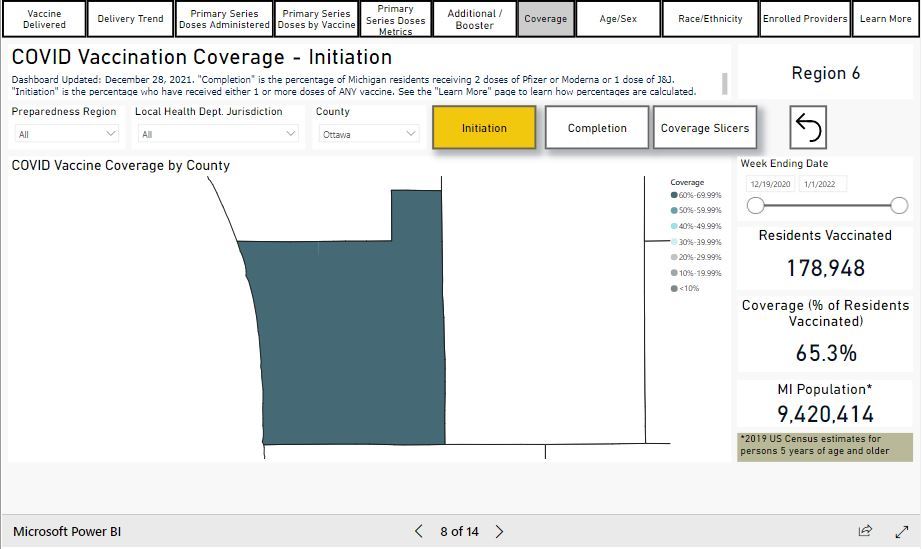 Coverage: 64.9% of all Ottawa County residents aged 5 years or older have received their first dose of the COVID-19 vaccine, as of December 27, 2021. More vaccine data from MDHHS can be found here.

Michigan Vaccination Rates
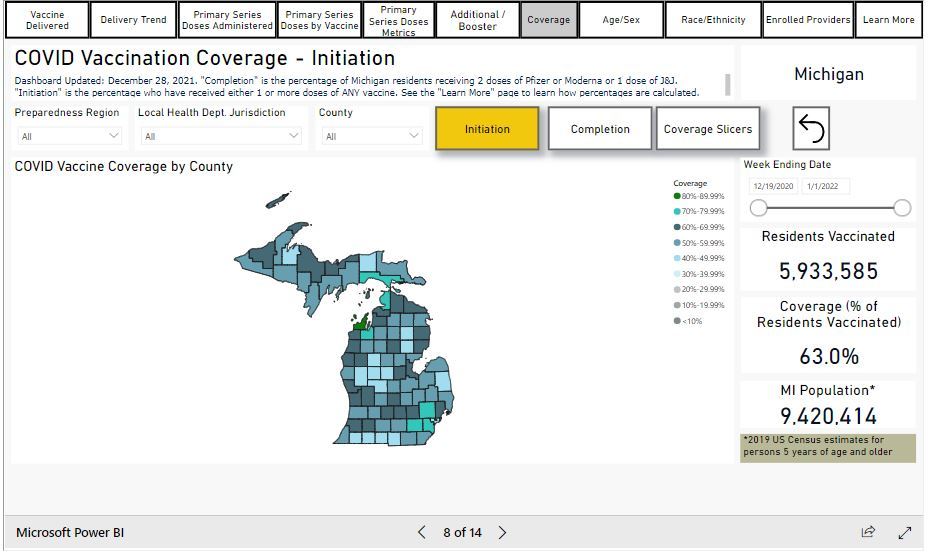 Coverage: 63.0% of all Michigan residents aged 5 years or older have received at least one dose of a COVID-19 vaccine, as of December 29, 2021. More vaccine data from MDHHS can be found here.
Progress is based on the CDC data tracker, which includes Michigan residents vaccinated by providers not currently reporting to the state dashboard below: Veterans Affairs, Department of Defense, Bureau of Prisons, and most out-of-state providers. Data provided in the Michigan COVID-19 Dashboard slightly undercounts the true number of doses administered to Michigan residents. LEARN MORE
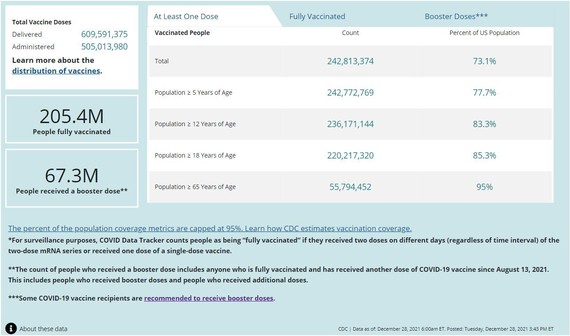
COVID-19 Vaccinations in the United States
Where to Find COVID-19 Vaccines
The OCDPH is listing all of its COVID-19 vaccination clinics on VaccinateWestMi.com. Click on the calendar to find scheduling and walk-in opportunities in Ottawa County and the surrounding area.
- The Pfizer two-dose vaccine is available for individuals aged 5 years or older.
- The Moderna two-dose vaccine is available for individuals aged 18 years or older.
- The Johnson & Johnson one-dose vaccine is available for individuals aged 18 years or older.
- Proof of date of birth is required (driver’s license, state ID or birth certificate). Parental consent is required for minors. Find more details and requirements on each clinic's page.
- Learn more about COVID-19 vaccines for children and teens.
 The Macatawa Area Express (MAX) and Allegan County Transportation are offering free rides for residents to vaccination appointments. To schedule a trip, call MAX at 616-355-1010. LEARN MORE - Kent County Vaccine Transportation Access Information HERE.
COVID-19 Testing
 Need Testing? No-Cost Community Sites. Rapid antigen testing for anyone with or without symptoms (parental consent required for minors). Samples are taken by nasal swab. Results within 15-30 min. No appointment needed • No doctor’s order needed • No insurance needed • No fee • Please bring a form of ID • No pets allowed • Wear a face covering. Find locations, dates and times at www.miOttawa.org/covid19
MDHHS Resources
Vaccine Information I MI Vaccine Locations I FAQs

More COVID-19 Vaccine Information Resources
VaccinateWestMi.com FAQs I Deaf & Hard of Hearing FAQ Videos
CDC COVID-19 FAQs Flyer I CDC FAQ Website
Alternative Languages: Arabic | Spanish | Korean | Russian | Simplified Chinese | Tagalog | Traditional Chinese | Vietnamese
Click to learn more from the CDC's COVID-19 Webinars and Partner Calls Videos
The Ottawa County Department of Public Health (OCDPH) sent this weekly update to those who signed up for vaccine notifications or COVID-19 updates. You may also sign up to receive information about other news topics from Ottawa County Parks, Sheriff’s Office and more. See the end of this email to make any changes to your subscription preferences.
|