|
Dear Immunization Partners,
The Michigan Department of Health and Human Services (MDHHS) Division of Immunization has received several reports of vaccine providers who have inadvertently administered Respiratory Syncytial Virus (RSV) vaccines (Arexvy or Abrysvo) to infants, rather than giving them the RSV monoclonal antibody product recommended for infants. The recommended monoclonal antibody product for infants is nirsevimab (trade name: Beyfortus). In addition, there are also reports of vaccine providers who have mistakenly administered Arexvy instead of Abrysvo to pregnant people.
MDHHS would like to review with all providers the proper use of the three RSV products available at this time. Below we listed what product is used for which population.
Adult RSV Products:
-
Abrysvo manufactured by Pfizer:
- For use in adults 60 years and older as a shared clinical decision-making conversation between patients and providers
-
For use in pregnant people 32 through 36 weeks pregnant during the RSV season of September through January to help prevent RSV in newborns
- Abrysvo is currently the only U.S. licensed RSV vaccine for pregnant people
- NOT TO BE USED FOR INFANTS/CHILDREN
-
Arexvy manufactured by GSK:
- For use in adults 60 years and older as a shared clinical decision-making conversation between patients and providers
- NOT TO BE USED FOR PREGNANT PEOPLE, INFANTS/CHILDREN
Pediatric RSV Product (monoclonal antibody):
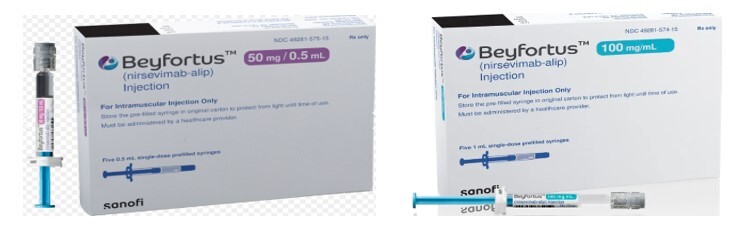 Suggestions for Preventing Administration Errors:
- If you carry more than one RSV product, keep them in separate areas of your storage unit
- Prominently label the different products and dose volumes (maybe use a different color of signage)
- Put age indications on the labels of these products in your storage unit
- Triple check the vaccine being given to make sure it is the correct vaccine for your patient
- Know your vaccines and who they are recommended for(e.g., age groups, medical status)
Resources:
If a vaccine administration error is made please file a Vaccine Adverse Event Reporting System (VAERS) report and feel free to reach out to checcimms@michgan.gov for further guidance.
Thank you for all you do.
Your Immunization Outreach and Education Team,
Alyssa, Andrea, Dianne, Heidi, Lisa, and Sarah
|