COVID-19 Vaccination Dashboard
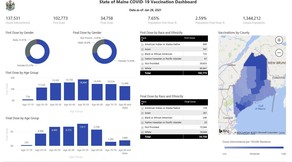 The State of Maine COVID-19 website now has a new interactive dashboard! The dashboard includes doses administered statewide, including vaccine supplied directly by the State to various locations and a long-term care vaccination program operated and supplied by the federal government.
Interim Clinical Considerations for Use of mRNA COVID-19 Vaccines Currently Authorized in the United States
Summary of recent changes:
- U.S. CDC continues to recommend that people get their second dose of COVID-19 vaccine as close to the recommended interval as possible (3 weeks for Pfizer-BioNTech, and one month for Moderna).
- U.S. CDC’s updated guidance was revised to allow for second dose administration up to 6 weeks (42 days) after the first if it is not feasible to adhere to the recommended interval. CDC is not advocating for people to delay getting their second dose, but the data from clinical trials support this range.
- U.S. CDC also updated guidance to allow that in exceptional situations, like when the first-dose vaccine product cannot be determined, any available mRNA COVID-19 vaccine may be administered at a minimum interval of 28 days between doses to complete the mRNA COVID-19 vaccination series.
- COVID-19 vaccines are not interchangeable. In extremely rare situations, some people simply may not have documentation or know which vaccine they got for their first dose.
For full list of changes, please visit the U.S. CDC website .
CDC launches two COVID-19 vaccination communication toolkits for community-based organizations and essential workers
This toolkit is designed for staff of organizations serving communities affected by COVID-19. These CBOs could include social service organizations, faith-based organizations, YMCAs/YWCAs, school organizations (e.g., PTAs/PTOs), meal delivery services, senior centers, and others. |
New COVID-19 Vaccine Web Pages for Pfizer-BioNTech and Moderna Vaccines
CDC has released two new web pages providing information on Pfizer-BioNTech and Moderna COVID-19 vaccines. The pages provide information on storage, handling, and administration information as well as useful tools for health care providers such as pre-vaccination checklists, standing order templates, storage unit temperature tracking logs and more. Also visit the general COVID-19 Vaccination page for overall clinical resources related to COVID-19 vaccine, including training, safety and requirements for vaccination providers participating in the program. Updates to ImmPact
Updates were released to ImmPact on January 25th, including the following:
Patients - Fields are required for race, ethnicity and address.
Providers may bypass this requirement by using the patient search under 'Immunizations' rather than patient search under 'Patients'.
Inventory - Only state users can Add Inventory; non-state users can add extra doses of COVID-19 vaccine via 'Modify Quantity'.
ImmPact Training for
COVID-19 Vaccine Sites
Trainings will take place every Wednesday, Thursday and Friday at noon through June 2021.
The agenda includes:
- New User Log in
- User Resources - forms, User Manual, Links
- Ordering COVID-19 vaccine through the Pandemic Module
- Accepting vaccine on arrival
- Patient search - entering immunization on patient to deduct from inventory vs. historical
- Reconciling inventory - modifying quantity
- Transferring vaccine to another site in ImmPact
- Error mitigation for sites sending through data exchange
Meeting link
Meeting ID: 859 5517 6464
Passcode: wi3gYq
Watch virtual ACIP meetings on February 24–25; no registration is required
Live, virtual meetings of the Advisory Committee on Immunization Practices (ACIP) will be held on February 24–25. The respective agendas (including times) are posted before the meetings and are expected to feature COVID-19 vaccination. No registration is required to watch the live ACIP meeting or listen via telephone. Look for instructions on how to sign up for public comment at the Public Comment tab of the ACIP web page.
|