|
Monitoring Monday – Quality Control Blanks
Join us Mondays as the Clean Water Team shares information and resources on watersheds and water quality monitoring. This Monday is about quality control blanks.

If you have spent time on a water quality monitoring field crew you have probably been asked to travel with or prepare some type of blank sample. When asking what these blanks are, you probably were told that they are used for quality control. And they are. Blank samples are one of the primary types of quality control samples used to evaluate and control the accuracy of field and laboratory analyses along with duplicates and spikes.
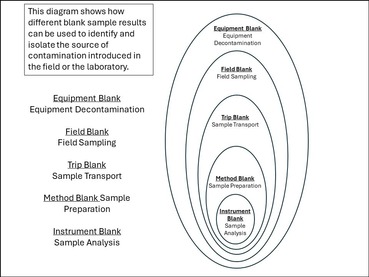
A blank is a solution that is prepared with water intended to be free of measurable concentrations of the analyte (a specific component measured in a chemical analysis) of interest. The source of contamination introduced in the field or laboratory can be deduced by comparing blank results. A value signal measured in a blank sample for an analyte that was absent in the blank solution is believed to be due to contamination.
Contamination may be due to sample containers, sampling equipment, filtration equipment, surroundings/ambient conditions, preservatives, transportation or storage practices, other samples, or laboratory analysis. So many types of blank samples are possible, each designed to segregate a different part of the data-collection process. An equipment blank could potentially be contaminated in the field, during transport to the lab or in the lab. The method blank, on the other hand, could only be contaminated in the lab. Using all blanks (appropriate for the project) described will facilitate the identification of contamination sources.
The frequency of using a specific type of blank and its corresponding analytes’ Measurement Quality Objective (MQO) should be specified in each project’s/program’s Quality Assurance Program Plan.
|
Ambient Blank - A sample of analyte free solution that is put in the same type of bottle/container used for an authentic environmental sample, kept with the set of sample bottles before sample collection, and opened at the site during sample collection and exposed to the ambient conditions.
Bottle Blank - A sample of analyte free media that is collected with the same bottle type as the environmental sample. Bottle blanks provide information about possible contaminants that may be introduced by sample bottles during collection. They are also used on a discretionary basis when new bottle types or lots are ordered for the first time.
Calibration Blank - An analyte free solution processed like any of the samples and used to "zero" the instrument. It is the first "sample" analyzed and used to set the meter to zero. and to confirm the absence of interferences in the analytical signal. It is analyzed with every set of samples that are extracted or digested. This reagent blank includes all reagents that are used in the analytical process and is carried through the entire process, including extraction and filtering or digestion to determine the contribution of bias from the reagents and analytical steps.
This is different from the field blank in that it is "sampled" in the lab. It is used to check the measuring instrument periodically for "drift" (the instrument should always read "0" when this blank is measured). It can also be compared to the field blank to pinpoint where contamination might have occurred. It is important to note that this blank is different than a field blank but can be used in combination with the field blank to help identify sources of contamination.
Equipment Blank/Instrument/Rinsate Blank – An analyte free solution that is processed through all of the sample equipment, typically during collection and processing of a sample, in a matter like the test sample or measurement. The most common method of collecting an equipment blank is to pour analyte free water over a recently decontaminated piece of equipment and collect the rinse water in a container that is appropriate for the analytical method that will be used. This blank is used to provide information about contaminants/bias that may be introduced during sample collection.
Field Blank – Probably the most common type of blank used and the most comprehensive as it is subjected to sample collection, transportation, preservation and storage, and laboratory analysis. These blanks consist of an analyte free solution consisting of a matrix comparable to the sample of interest. The field blank is taken to the sampling location, transferred to the appropriate container, preserved (if required by the method), and treated the same as the corresponding sample type during the course of a sampling event and shipped to the laboratory with field samples.
Filter Blank – An analyte free solution that is filtered in the same manner and through the same filter apparatus used for an environmental sample.
Fortified Method Blank - This type of method blank is spiked with analyte. Not to be confused with spike samples, used to determine analyte recovery, fortified method blanks are used to assess analyte degradation during analysis procedures.
Laboratory Blank - Sample matrix representative of the environmental sample (i.e., water, sand, etc.) that is prepared in the laboratory and is free from the analytes of interest. These samples are then analyzed alongside test (environmental) samples.
Matrix Blank - These blanks contain all sample components except the analytes of interest and are subject to all sample processing steps. They are used to measure the presence of significant interference from the matrix.
Method Blank – Method blanks are used to determine background contamination or interferences in the analytical system. The method blank is composed of the sample matrix, absent the analyte(s) of interest. These blanks are analyzed along with the test (environmental) samples to measure the response of the analytical system to the analyte at a theoretical concentration of zero.
Preservation Blank – An analyte free solution that is treated with the same preservatives used for an environmental sample. When compared with a blank without the preservative, the effect of the preservation agent can be determined.
Reagent Blank - A reagent blank is an analyte free solution (sometimes approximating the sample matrix) that undergoes all the same treatment as a test (environmental) sample. It is to verify that any positive response to your test sample is a result of the test sample itself and not any testing conditions including contaminated analytical reagents, solvents or glassware. If your reagent blank has a positive response the same as what you are looking for, then the whole batch of analysis is potentially contaminated and needs to be repeated.
Sample Blank – A sample blank refers to using an analyte free solution for zeroing an instrument during a test procedure.
Sampler Blank – An analyte free solution that a field technician pours or pumps through the same field sampler used for collecting a test (environmental) sample.
Splitter Blank – An analyte free solution that is mixed and separated using a field splitter in the same manner and through the same apparatus used for a test (environmental) sample.
Source Solution Blank - A sample of analyte free media that is transferred to a sample bottle in an area of the office/laboratory in a clean and protected atmosphere with respect to target analytes.
Temperature Blank – A sample container containing analyte free solution and transported with each shipment of collected samples to determine the temperature of other samples in the shipment on arrival at a laboratory. The purpose of a temperature blank provides a fair representation of sample temperature upon laboratory receipt.
Transfer Blank – Transfer blanks are prepared in the field while splitting or transferring samples. They may indicate contamination from sampling equipment, the surroundings, sample containers, or cross-contamination during shipment. Each transfer blank requires the use of two pre-cleaned sample containers. One container is filled at the laboratory with analyte free solution and serves as the source for the transfer. The second container is sent empty and serves as the receiving container; this container is the transfer blank to be analyzed. Transfer blank water (analyte free solution/matrix) must be from the same source as the method blank water (analyte free solution/matrix ) used by the laboratory performing the analysis. Transfer blanks are prepared in the most contaminated sampling area in an attempt to simulate a worst-case scenario regarding environmental contributions to sample contamination.
Trip/Transport/Travel Blank – An analyte free solution that is taken from the laboratory to the sampling site and transported back to the laboratory without having been exposed to sampling procedures. They may indicate contamination from the sample containers, cross-contamination during shipment, storage, or laboratory contamination, and are especially valuable for volatile compounds and those subject to degradation by factors such as exposure to light, bacteria, or freeze–thaw cycles.
|

To ensure minimal contamination, obtain, distilled or deionized water is often used for preparing blanks, but not all laboratory grade waters are equal. To ensure that your prepared blank will be adequate for your quality assurance and quality control needs, the proper laboratory grade water to prepare the blank must first be chosen.
Reagent-grade Water - The term “reagent-grade” is used to designate chemical substances that meet the purity standards required to ensure the precision and reliability of chemical reactions, chemical analysis, and/or physical testing. Purity standards are specified by the American Chemical Society, ASTM International, ISO, and other similar organizations. The production of ultrapure is the result of the combination of various water purification technologies which are as follows: synthetic activated carbon method, ion-exchange resin method, microfiltration, UV-photo-oxidation lamps, and ultrafiltration membranes.
Distilled Water - Distillation is a process used to demineralize water. In distillation, water is boiled, and the steam is collected and then cooled to return it to its liquid state. Salt, particulates, and most other impurities are left behind as the water turns to vapor, but some impurities, such as volatile organics and mercury, will vaporize along with the water.
Deionized Water - Deionized water is purified by running water through a form of electrically charged resin, usually a mixed ion exchange bed containing both positive and negative charged resins. Deionization does not remove molecular contaminants (such as salt) or uncharged organic particles like bacteria or viruses. Deionized water is reactive, and its properties, particularly its pH level, will change once it is exposed to air.
Reverse Osmosis (RO) Water - Reverse osmosis (RO) is a made through a purification process that uses a semi-permeable membrane to separate water molecules from other substances. RO can remove dissolved or suspended chemical species as well as biological substances (principally bacteria). It is used in industrial processes and the production of potable water.
Type I Water: Ultrapure Water- Type I water is the purest form of water and is often referred to as ultrapure water. Due to its pure state, Type I water is ideal for procedures that are highly sensitive procedure which are susceptible to contamination. To make achieve ultrapure water, it often undergoes ultra-pure polishing which can be carried out using ultra-pure water systems.
- Resistivity: Greater than 18 MΩ-cm ; Conductivity: Less than 0.056 µS/cm; Total organic carbons: Less than 50 ppb
Type II Water: Purified Water – Used in general lab use. Type II water has a high level of purity and is often used for standard applications, tests, and procedures. This type of water can be produced through reverse osmosis and ion exchange which involves the removal of synthetic resins, which are replaced by hydrogen or hydroxyl ions. Type II water can also be produced through electrical ion exchange, which uses active purification technology to combine electrodialysis with ion-exchange, or distillation.
- Resistivity: Greater than 1 MΩ-cm; Conductivity: Less than 1 µS/cm; Total organic carbons: Less than 50 ppb
Type III Water: Primary Grade Water – This type of water is produced through purification processes such as media filter, carbon filter and reverse osmosis, which is a water purification process that removes approximately 90 – 99% of contaminants from standard tap water. This water is used for non-critical work like rinsing beakers.
- Resistivity: Greater than 4 MΩ-cm; Conductivity: Less than 0.25 µS/cm; Total organic carbons: Less than 200 ppb
Type IV Water: Most often used as a feed to create more pure water, Type IV water is produced through reverse osmosis
- Resistivity: 200 KΩ-cm; Conductivity: Less than 5 µS/cm; Total organic carbons: No standard
|
RESOURCES & REFERENCES:
Clean Water Team - CWT - Quality Assurance/Quality Control (QA/QC) https://www.waterboards.ca.gov/water_issues/programs/swamp/clean_water_team/technical-support.html
Design, Analysis, and Interpretation of Field Quality-Control Data for Water-Sampling Projects https://pubs.usgs.gov/tm/04/c04/
Different Types of Pure Water For The Lab: What You Need to Know https://www.elgalabwater.com/blog/different-types-pure-water-lab-what-you-need-know
Design, Analysis, and Interpretation of Field Quality-Control Data for Water-Sampling Projects https://pubs.usgs.gov/tm/04/c04/
Fact Sheet: Quality Control Tools: Blanks. April 27, 2009. EPA Region 3 QA Staff https://19january2017snapshot.epa.gov/quality/quality-control-tools-factsheet-blanks_.html
High Purity Water: A Technical Guide to Types I, II & III https://www.evoqua.com/siteassets/documents/products/disinfection/ds-lab-techguide-wp-0522.pdf
Laboratory Water: Its Importance and Application https://orf.od.nih.gov/TechnicalResources/Documents/DTR White Papers/Laboratory Water-Its Importance and Application-March-2013_508.pdf
National field manual for the collection of water-quality data: U.S. Geological Survey Techniques of Water-Resources Investigations https://water.usgs.gov/owq/FieldManual/index.html
Q & A: What are “Blanks”? - Watermarks: The California Newsletter for Citizen Water Quality Monitoring, Issue 15 https://www.waterboards.ca.gov/water_issues/programs/swamp/docs/cwt/newsletter/wm_sf_2014.pdf
Quality Assurance, Quality Control, and Quality Assessment Measures (USEPA) https://archive.epa.gov/water/archive/web/html/132.html
Quality-Control Design for Surface-Water Sampling in the National Water-Quality Network (USGS) https://pubs.usgs.gov/of/2018/1018/ofr20181018.pdf
Quality-Control of Water-Quality Samples (USGS) https://pubs.usgs.gov/wdr/2004/wdr-il-04/misc/qcofples.htm
Quality-control design for surface-water sampling in the National Water-Quality Assessment Program: U.S. Geological Survey Open-File Report 1997–223 https://pubs.usgs.gov/of/1997/223/index.html
Reverse Osmosis https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/reverse-osmosis
Surface Water Ambient Monitoring Program Quality Assurance Program Plan https://www.waterboards.ca.gov/water_issues/programs/swamp/docs/swamp-qaprp-2022.pdf
SWAMP - Quality Assurance https://www.waterboards.ca.gov/water_issues/programs/swamp/quality_assurance.html
The Volunteer Monitor's Guide to Quality Assurance Project Plans https://www.epa.gov/sites/default/files/2015-06/documents/vol_qapp.pdf
Ultra-Pure vs Feed Water, Comparing the 4 Types of Laboratory Water https://www.technologynetworks.com/immunology/lists/4-types-of-laboratory-water-made-simple-293547
What’s the difference Between Type I, II, III & IV Water? https://www.labconco.com/articles/water-type-difference
What Water Type Should I use? https://www.labconco.com/articles/water-type-difference
|