COVID-19 Alaska Clinical Update
Wednesday, September 16, 2021
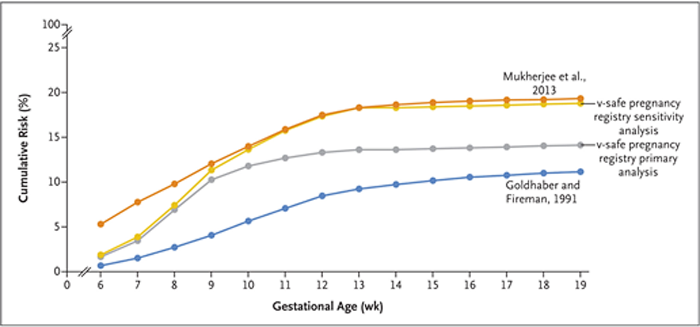
Risk of miscarriage is similar in unvaccinated women and women who received mRNA COVID vaccine
In a JAMA research letter published on September 8, researchers analyzed the odds of receiving a COVID-19 vaccine in the 28 days prior to spontaneous abortion compared with the odds of receiving a COVID-19 vaccine in the 28 days prior to index dates for ongoing pregnancies. Data was collected from the Vaccine Safety Datalink, a collaboration between the CDC and 9 health systems that represents approximately 3% of the US population. Of 105,446 unique pregnancies, 13,160 spontaneous abortions and 92,286 ongoing pregnancies were identified. Overall, 7.8% of women received 1 or more Pfizer-BioNTech vaccines; 6.0% received 1 or more Moderna vaccines; and 0.5% received a Janssen vaccine during pregnancy and before 20 weeks’ gestation. Spontaneous abortions did not have an increased odds of exposure to a COVID-19 vaccination in the prior 28 days compared with ongoing pregnancies (adjusted odds ratio, 1.02; 95% CI, 0.96-1.08).
Risk of miscarriage is similar in vaccinated women and historical comparator cohorts
In a NEJM letter to the editor published on September 8, authors analyzed data from the CDC’s v-safe Covid-19 vaccine pregnancy registry to determine the cumulative risk of spontaneous abortion from 6 to less than 20 weeks of gestation. 2456 participants with a singleton pregnancy who had received at least one dose of an mRNA Covid-19 vaccine either before conception or before 20 weeks of gestation and who did not have a spontaneous abortion before 6 weeks of gestation were included. The cumulative risk of spontaneous abortion from 6 to less than 20 weeks of gestation was 14.1 in the primary analysis and 12.8% in an analysis using direct maternal age–standardization to the reference population. As compared with data from two historical cohorts that represent the lower and upper ranges of spontaneous-abortion risk, the cumulative risks of spontaneous abortion from our primary and sensitivity analyses were within the expected risk range.
Crisis Standards of Care in Idaho
Hospitals in northern Idaho began operating under “crisis standards of care,” allowing healthcare workers to ration care as facilities struggle to handle an influx of COVID-19 patients amid an increase in cases and staff shortages. This decision was enacted by the Idaho Department of Health and Welfare and applies to 10 hospitals and healthcare systems in the panhandle and north-central regions. It allows hospitals to apportion certain resources, such as ICU beds, to patients they deem most likely to survive. Other patients will still receive care but might go without some life-saving medical equipment.
On Tuesday, September 21 from 7-8pm there is an ECHO for healthcare providers in Alaska to discuss providing care for patients with limited resources.
Persons who have recovered from SARS-CoV-2 infection and were subsequently vaccinated have robust antibody protection against multiple variants in laboratory study
In this pre-print study, researchers evaluated the in-vitro antibody response to polymutant spike proteins from the plasma of individuals who had been infected and recovered from SARS-CoV-2 and then subsequently received an mRNA vaccine. First, researchers created polymutant spike proteins that were an amalgamation of various mutations that have occurred across different variants of concern. They found that 20 naturally occurring mutations in the SARS-CoV-2 spike protein were enough to confer almost complete resistance to the neutralizing antibodies produced independently by either persons who had recovered from SARS-CoV-2 infection or individuals who had only received a COVID-19 mRNA vaccine. Plasma from individuals who had been infected and subsequently received mRNA vaccination, neutralized this highly resistant SARS-CoV-2 polymutant spike protein and at least five other related viruses, such as SARS-CoV-1 and viruses found in bats and pangolins.
*This article has not gone through peer review and is in pre-print status. It may have not been finalized by authors, might contain errors, and report information has not yet been accepted or endorsed in any way by the scientific or medical community.
Child cases of COVID-19 increasing exponentially
According to data from the American Academy of Pediatrics released on September 9, more than 243,000 cases of COVID-19 in children were reported in the past week, the second highest number of child cases in a week since the pandemic began. After declining in early summer, child cases have increased exponentially with nearly 500,000 cases in the past 2 weeks.
More than 83% of blood donations in May 2021 contained either infection-induced or vaccine-induced COVID-19 antibodies
In this repeated cross-sectional study of blood donation specimens, researchers tracked the seroprevalence of SARS-CoV-2 antibodies in donated blood products from July 2020 to May 2021. The study included more than 1.4 million specimens from a catchment area that represents 74% of the US population. Estimated SARS-CoV-2 seroprevalence weighted for differences between the study sample and general population increased from 3.5% in July 2020 to 20.2% for infection-induced antibodies and 83.3% for combined infection- and vaccine-induced antibodies in May 2021. Infection-induced SARS-CoV-2 seroprevalence was consistently highest in Hispanic and non-Hispanic Black persons, mirroring case trends. However, by May 2021, non-Hispanic Asian and White persons had significantly higher combined seroprevalence estimates, likely because non-Hispanic Asian and White persons received vaccinations at a greater rate than Hispanic and non-Hispanic Black persons. Seroprevalence estimates indicate that the number of COVID-19 cases reported are fewer than the number of SARS-CoV-2 infections that have occurred, likely because many mildly symptomatic and asymptomatic individuals did not seek testing or clinical care. The data in this study suggest that for every COVID-19 case reported, the number of people infected and with detectable SARS-CoV-2 antibodies decreased from 3.1 in July 2020 to 2.1 in May 2021.
Expanded federal COVID-19 vaccine mandate covers nearly 100 million Americans
On September 9, President Biden announced an expanded vaccine mandate that will requires all private sector companies employing more than 100 people to mandate COVID-19 vaccination or conduct weekly testing, affecting about 80 million people. Workers at healthcare facilities that receive Medicare or Medicaid funding, about 17 million people, also will have to be vaccinated, extending an earlier requirement for workers at nursing homes to include facilities such as hospitals, home-health agencies, and dialysis centers. President Biden also is requiring all executive branch employees and federal contractors to be fully vaccinated, with no testing option, covering several million more workers. Additionally, employees of Head Start programs and schools run by the Department of Defense and Bureau of Indian Education, about 300,000 people, will be required to be vaccinated. It is still uncertain when this mandate would go into effect and what the enforcement mechanism would be.
CDC Morbidity and Mortality Weekly Reports (MMWR):
Full vaccination reduces risk of infection five-fold, risk of hospitalization ten-fold, and risk of death by eleven-fold
Full vaccination reduces risk of infection five-fold, risk of hospitalization ten-fold, and risk of death by eleven-fold
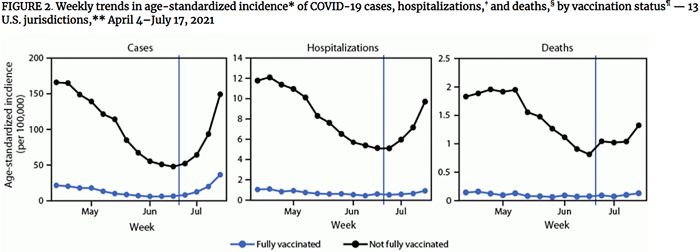
In this MMWR, researchers used data from 13 public health jurisdictions around the US to compare calculated age-standardized ratios of cases, hospitalizations, and deaths between fully vaccinated and unvaccinated persons from April 4-June 19, 2021 (pre-Delta) and June 20-July 17 (post-Delta). Reporting results in terms of incident rate ratios, authors found that risk of infection among those fully vaccinated was 11.1x lower than among unvaccinated persons for the pre-Delta period and for the post-Delta period, the risk of infection for fully vaccinated persons was only 4.6x lower than for unvaccinated persons. Compared to the unvaccinated, the risk of hospitalization for fully vaccinated persons was 13.3x lower and 10.4x lower in pre- and post-Delta periods respectively. The risk of death for fully vaccinated persons was 16.6x and 11.3x lower for the pre- and post-Delta periods. By incorporating data on vaccine rates during these two periods, authors were able to calculate estimates for vaccine efficacy against infection, hospitalization, and death. For vaccine protection against infection, they estimated that effectiveness dropped from 91% to 78% with Delta. For protection against hospitalizations, effectiveness appeared to go from 92% to 90%, and for death, from 94% to 91%.

Combined vaccine efficacy against COVID-19 hospitalization is 86% for all ages in months when Delta variant was most prevalent; efficacy in adults aged ≥75 years significantly lower than for younger adults
Using data from the VISION network of 187 hospitals and 221 emergency departments and urgent care clinics (ED/UC) from June-August 2021, authors assess the real-world efficacy of the 3 authorized COVID-19 vaccines against a COVID-19 associated ED/UC visit or hospitalization in the months when the Delta variant was predominant. Overall, for 14,636 adults hospitalized for COVID-19, vaccine efficacy against COVID-19 hospitalization was 86%. Efficacy was significantly lower among adults aged ≥75 years (76%) than among those aged 18–74 years (89%). Across all ages, efficacy against hospitalization was significantly higher among Moderna vaccine recipients (95%) than among Pfizer-BioNTech (80%) or Janssen (60%) vaccine recipients. Among 18,231 adults with COVID-19-related ED/UC visits, vaccine efficacy against ED/UC encounters was 82%. Efficacy was highest among Moderna vaccine recipients (92%), followed by Pfizer-BioNTech vaccine recipients (77%), and was lowest (65%) for Janssen vaccine recipients.
mRNA vaccines highly effective at preventing COVID-19 hospitalization in vaccinated US Veterans; lower efficacy seen in adults aged ≥65 years
This study examined the hospitalization of 1,175 U.S. veterans aged ≥18 years at five Veterans Affairs Medical Centers (VAMCs) during February 1–August 6, 2021 to evaluate the efficacy of mRNA COVID vaccines. Overall adjusted vaccine effectiveness against COVID-19–associated hospitalization was 86.8% and was similar before (February 1–June 30) and during (July 1–August 6) SARS-CoV-2 Delta variant predominance. Vaccine effectiveness was 79.8% among adults aged ≥65 years and 95.1% among those aged 18–64 years. Adjusted vaccine effectiveness estimates were not statistically different for Pfizer-BioNTech (83.4%) and Moderna vaccines (91.6%) with overlapping 95% confidence intervals.
Close contacts who had exposure to an unmasked colleague had higher odds of a positive test result; each additional exposure was associated with a 40% increase in odds of a positive test
In January 2021, Saint Louis University implemented a modified quarantine protocol that considered mask use when determining which close contacts required quarantine. During January–May 2021, 265 students received a positive SARS-CoV-2 test result; these students named 378 close contacts. Compared with close contacts whose exposure only occurred when both persons were masked (7.7%), close contacts with any unmasked exposure (32.4%) had 4-fold higher risk of receiving a positive SARS-CoV-2 test result (adjusted odds ratio = 4.9). Any additional exposures were associated with a 40.0% increase in odds of a positive test result (adjusted odds ratio = 1.4). Notably, this study occurred before the Delta variant became the predominant strain, which is nearly 50% more transmissible than prior strains.
RECURRENT TOPICS
Providing COVID-19 vaccinations
We recommend that all healthcare providers outreach to their patients who are immunocompromised and are candidates for a third vaccine dose. A third dose of mRNA COVID vaccine is recommended in certain immunocompromised individuals, specifically solid organ transplant recipients or those who are diagnosed with conditions that are considered to have an equivalent level of immunocompromise (active lymphoma/leukemia treatment, stem cell transplant recipient, active treatment with high-dose corticosteroids).
If you are interested in providing the COVID-19 vaccine in your office or clinic, please visit the COVID-19 Vaccination Program Provider Enrollment page. If you have additional questions, please email Matthew Bobo at matthew.bobo@alaska.gov
Monoclonal Antibodies
Monoclonal antibody treatment for COVID-19, REGEN-COV (casirivimab and imdevimab), has been approved to treat mild-moderate COVID-19 and for post-exposure prophylaxis of COVID-19 in individuals age 12 years and older who are at high risk for progression to severe COVID-19.
If you are interested in providing monoclonal antibody therapy for COVID-19 in your office or clinic, please refer to this guide from the U.S. DHSS, and then send an email to Coleman Cutchins (coleman.cutchins@alaska.gov) and CJ Kim (cj.kim@alaska.gov) for local assistance.
For the latest recommendations, check out the CDC webpage on Monoclonal Antibodies for High-Risk COVID-19 patients and COVID-19 Monoclonal Antibody Resources for Healthcare Providers.
Ivermectin
On August 26, the CDC issued a Health Advisory about the increase in ivermectin prescriptions during the 2021 summer and an associated rise in the number of calls to poison centers reporting overdoses or adverse effects.
According to the Merck, the drug company that manufactures ivermectin, there is:
- No scientific basis for a potential therapeutic effect against COVID-19 from pre-clinical studies;
- No meaningful evidence for clinical activity or clinical efficacy in patients with COVID-19 disease, and;
- A concerning lack of safety data in the majority of studies.
A Cochrane Review published July 28 concluded, the “reliable evidence does not support the use of ivermectin for treatment or prevention of COVID‐19.” In addition, the FDA has recently created a webpage further explaining why you should not use ivermectin to treat or prevent COVID-19 and the potential harms of taking a veterinary formulation of this mediation. Infectious Disease Society of America (IDSA) guidelines do not recommend ivermectin.
Post-acute Sequelae of COVID-19 (PASC)
For the latest recommendations, check out the CDC webpage on Post-COVID-19 Syndrome and Evaluating and Caring for Patients with Post-COVID conditions
Myocarditis
For the latest recommendations, check out the CDC webpage on myocarditis and COVID-19 vaccines
COVID-19 Speakers’ Bureau
Anyone can request a free presentation for a group interested in learning more about the COVID-19 vaccines available in Alaska.
Aside from COVID-19
CDC and ACIP release recommendations for seasonal Influenza vaccination
The 2021-2022 influenza season is expected to arrive late fall through early spring. Routine annual influenza vaccination is recommended for all persons aged ≥6 months who do not have contraindications. Detailed recommendations for influenza vaccination for persons currently infected with COVID-19 are available from CDC. Influenza vaccine may be administered at the same time as COVID-19 vaccine.
Upcoming Events/Conferences/Presentations
Alaska Maternal Child Health and Immunization Conference is accepting abstracts
The MCH&I Conference, hosted by Alaska Native Tribal Health Consortium’s Alaska Native Epidemiology Center and the Alaska Dept. of Health & Social Services, Division of Public Health, Section of Women’s, Children’s, and Family Health, is accepting abstracts until Friday, September 17, 2021. There is still time to submit your abstracts by clicking the link - https://www.surveymonkey.com/r/MCHIAbstracts.
Application deadline for Northwest Public Health & Primary Care Leadership Institute approaching on October 1, 2021
Applications for the 2022 Northwest Public Health & Primary Care Leadership Institute are due October 1, 2021 and some partial scholarship funds are available. The Leadership Institute is an offering from the Northwest Center for Public Health Practice and the Northwest Regional Primary Care Association. The program builds on the long-standing training programs of our collaborating organizations and is designed to help mid-career public health and primary care professionals become the next generation of leaders in their fields. The 9-month program runs from January - September 2022, and will develop collaborative, adaptable leaders who can work effectively within and across fields to improve community health.
ANTHC Tribal Health Webinar Series
The ANTHC Tribal Health Webinar series occurs on Friday from 12-1pm on Zoom and is open to the public. Here is the upcoming schedule for the fall and the Zoom link.
- 09/17: Rebecca Robinson, PhD: ANTHC Behavioral Health Wellness Clinic
- 09/24: Jason Capo, MD and Ben Westley, MD: ANMC Septic Arthritis Guideline
- 10/01: Michelle Rothoff, MD: TB in Alaska 101
- 10/08: Rodrick Smith, MD: Diagnosis and Management of Childhood Seizures
- 10/22: Elisha Brownson, MD: Imaging Decisions in Pediatric Trauma
- 11/05: Rosalyn Singleton, MD: Respiratory/RSV/COVID Hospitalization trends and future interventions.
- 11/15: Mary Owen, MD: Tlingit People
- 11/19: Katie Presser, PharmD: Statewide Antimicrobial Stewardship
Join Zoom Meeting
https://anthc.zoom.us/j/98667611681
Meeting ID: 986 6761 1681
One tap mobile
+16699006833,,98667611681#
Free opioid prescribing education on October 26
On October 26 from 11am-2pm, the Division of Behavioral Health has partnered with Boston University School of Medicine to provide a free opioid prescribing education opportunity. This workshop is intended for physicians, nurse practitioners, registered nurses, physician assistants, nurses, dentists, pharmacists, and allied health professionals who manage acute and chronic pain. SCOPE of Pain is a series of continuing medical education/continuing nursing education activities designed to help you safely and effectively manage patients with acute and/or chronic pain, when appropriate, with opioid analgesics.
- Effective communication skills and the potential risks and benefits of opioids for managing acute and chronic pain
- The assessment of opioid misuse risk and recognition and treatment of opioid use disorder
- Helping you safely and effectively manage patients with acute and/or chronic pain, when appropriate, with opioid analgesics
Registration required at scopeofpain.org (select core curriculum from the top menu, choose live conferences, select the red button)

Use of Valid Background Check Clearances by Multiple Employers
Providers who have applicants with pending background checks with DHSS BCU may allow individuals to start work prior to the Background Check Program completing their check IF the applicant has documented proof of having a current Alaska DHSS background check (provisional or complete) for another DHSS provider. The applicant must provide their new employer a copy of the final background check eligibility letter for the other provider, and providers are required to maintain a copy of that eligibly document in the employee’s file. Providers who hire employees in this manner MUST provide supervised access to those employees, i.e., to ensure the protection of recipients of services, the provider must maintain a prudent level of awareness of the whereabouts of an individual for whom supervised access is required. Employees working in this way may NOT be the only individual working in a facility or a setting, as another employee must be present to provide the supervised access. The provider’s oversight agency will monitor providers to ensure they are in compliance with these requirements, and providers must submit proof of compliance to those agencies upon request. Questions? Please contact your DHSS oversight division.
CDC Clinical Support: There is a Clinician On-Call Center, a 24-hour hotline with trained CDC clinicians standing by to answer COVID-19 questions. Call 1-800-CDC-INFO (800-232-4636) and ask for the Clinician On-Call Center.
All Alaskans and people who work or live in Alaska who are aged 12 years and older are eligible for vaccination against COVID-19. Appointments can be made at covidvax.alaska.gov.
The most up-to-date, evidence based COVID-19 treatment guidelines can be found at:
IDSA Guidelines on the Treatment and Management of Patient with COVID-19
NIH COVID-19 Treatment Guidelines
|