
Jessica Jenkins, Kristine Smith, Diana Riner
The Michigan Department of Health and Human Services (MDHHS) Bureau of Laboratories (BOL) provides arbovirus testing for the State of Michigan. The arbovirus panel at the BOL includes West Nile virus (WNV), St. Louis encephalitis virus (SLE), EEE, and California group virus- Lacrosse antigen (CGV). The screening test for arboviruses detects IgM antibodies in cerebrospinal fluid. Patient samples that are positive or inconclusive by the IgM test will undergo a confirmatory test known as the “plaque reduction neutralization test” or “PRNT”. As antibodies produced against one arbovirus can weakly bind to other arboviruses and “cross-react”, the combination of IgM testing and the confirmatory PRNT are often needed for confirmation of infection.
The Michigan Department of Health and Human Services (MDHHS) Bureau of Laboratories (BOL) provides arbovirus testing for the State of Michigan. The arbovirus panel at the BOL includes West Nile virus (WNV), St. Louis encephalitis virus (SLE), EEE, and California group virus- Lacrosse antigen (CGV). The screening test for arboviruses detects IgM antibodies in cerebrospinal fluid. Patient samples that are positive or inconclusive by the IgM test will undergo a confirmatory test known as the “plaque reduction neutralization test” or “PRNT”. As antibodies produced against one arbovirus can weakly bind to other arboviruses and “cross-react”, the combination of IgM testing and the confirmatory PRNT are often needed for confirmation of infection.
 |
 |
MDHHS Bureau of Laboratory Scientist
|
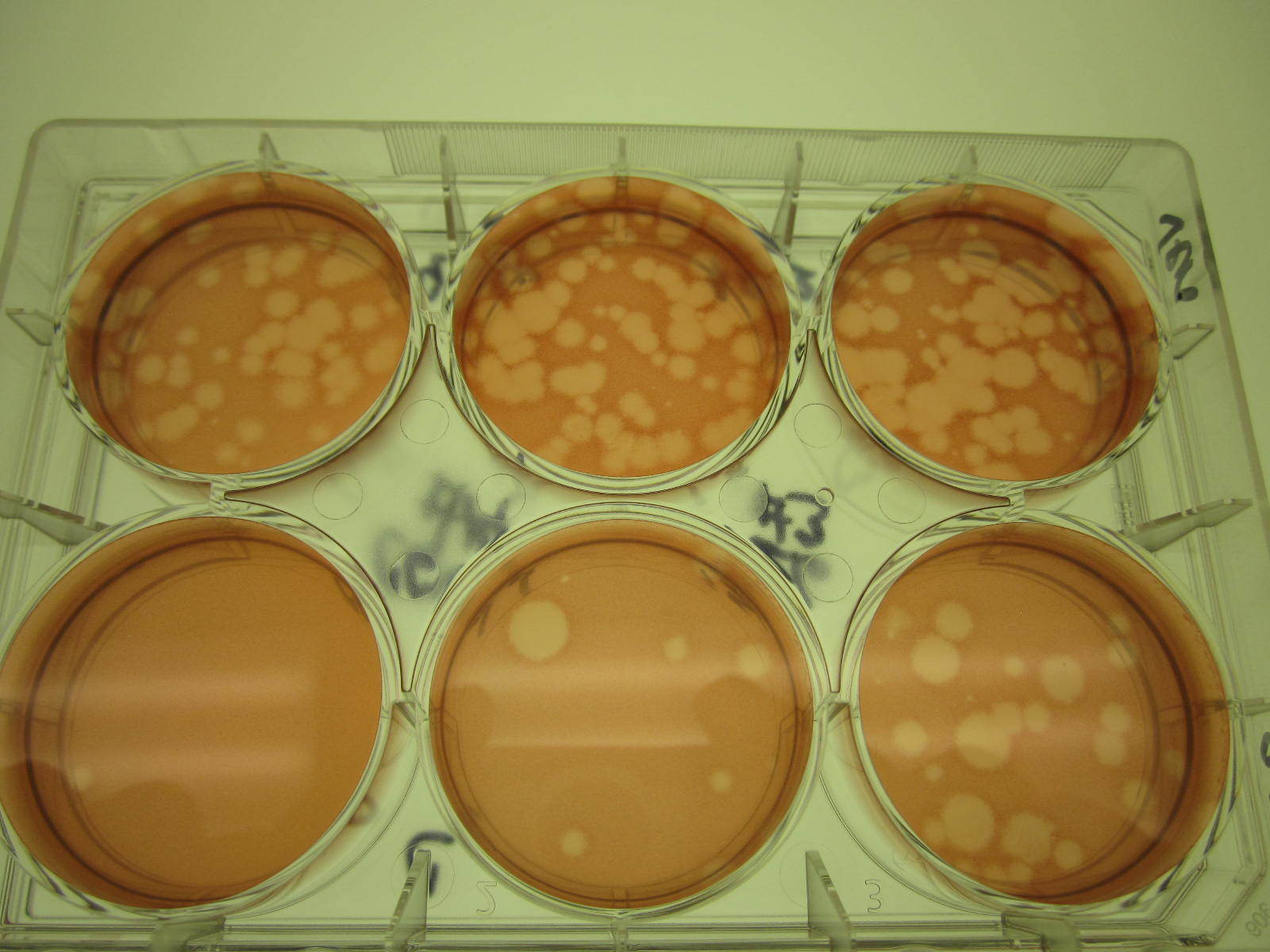
Plaques formed during PRNT
|
When people think of mosquito-borne diseases, West Nile virus is usually what comes to mind. Most people have never heard of a virus called Eastern equine encephalitis virus, usually referred to as “EEE” or “triple E”. Though it’s not as well known, EEE has been present in Michigan for decades. Per the CDC, human cases of neuroinvasive EEE are sporadic, with only seven cases reported in Michigan from 2009-2018. In 2019 however, ten human cases of EEE were reported in Michigan with six fatalities. Testing for EEE requires highly trained microbiologists and special laboratory space as arboviruses can cause severe disease in humans. Michigan is one of seven places in the US capable of performing EEE confirmatory testing.
|
MDHHS BOL scientist
|
PRNT is based on the ability of antibodies in the patient specimen to neutralize live virus. Specimens are incubated with the virus in question (WNV, SLE, EEE, or CGV) then added to a plate with cells. If the virus is not neutralized, it will infect the cells and cause the formation of plaques, which can be counted when the plate is placed on a lighted surface. At MDHHS, PRNT is performed to confirm EEE, SLE, WNV, CGV, Zika, and Dengue types 1 and 2 arboviral infections. As this test uses live virus, it requires a specialized biosafety level 3 (BSL-3) laboratory space. BSL-3 laboratories have enhanced air handling systems and extra security to ensure safety. In addition, microbiologists who work in the BSL-3 laboratory and perform this testing undergo additional safety training and wear extra personal protective equipment (PPE) including respirators. The capability to perform this specialized testing for Michigan residents during the recent EEE outbreak decreased turn-around-times for results, allowing for faster detection and diagnosis of disease. Maintaining BSL-3 testing capabilities allows for a rapid response to biological threats. In the case of EEE, the BOL was able to immediately begin confirmatory testing once the first cases screened positive for anti-EEE IgM antibodies. Without the designated BSL-3 laboratory space, specimens would have to be shipped out for testing which could delay results by several weeks. When viruses like EEE emerge as a public health threat, the ability to perform BSL-3 level testing is essential for providing laboratory confirmation of clinical infections.
|
||