|
COVID-19 Health Alert # 19:
Updated Recommendations from the US COVID-19 Treatment Guidelines Panel
Bureau of Communicable Diseases, October 15, 2020
Summary
-
The COVID-19 Treatment Guidelines Panel has released updated clinical guidance for the management of patients with COVID-19, which are now available online.
- The panel currently recommends pharmacologic therapy with remdesivir and/or dexamethasone for hospitalized patients who require supplemental oxygen.
- While clinical trials of additional treatment strategies are ongoing, the Panel has determined that there is currently insufficient evidence to recommend for or against the use COVID-19 convalescent plasma, SARS-CoV-2 immunoglobulins, or other adjunctive treatments.
Dear Colleagues,
To inform clinicians about the evolving scientific literature about COVID-19 therapeutic strategies and provide evidence-based treatment recommendations, the National Institutes of Health convened the COVID-19 Treatment Guidelines Panel. These guidelines have been periodically updated as new evidence accumulates, and are available for review at https://www.covid19treatmentguidelines.nih.gov/therapeutic-management/.
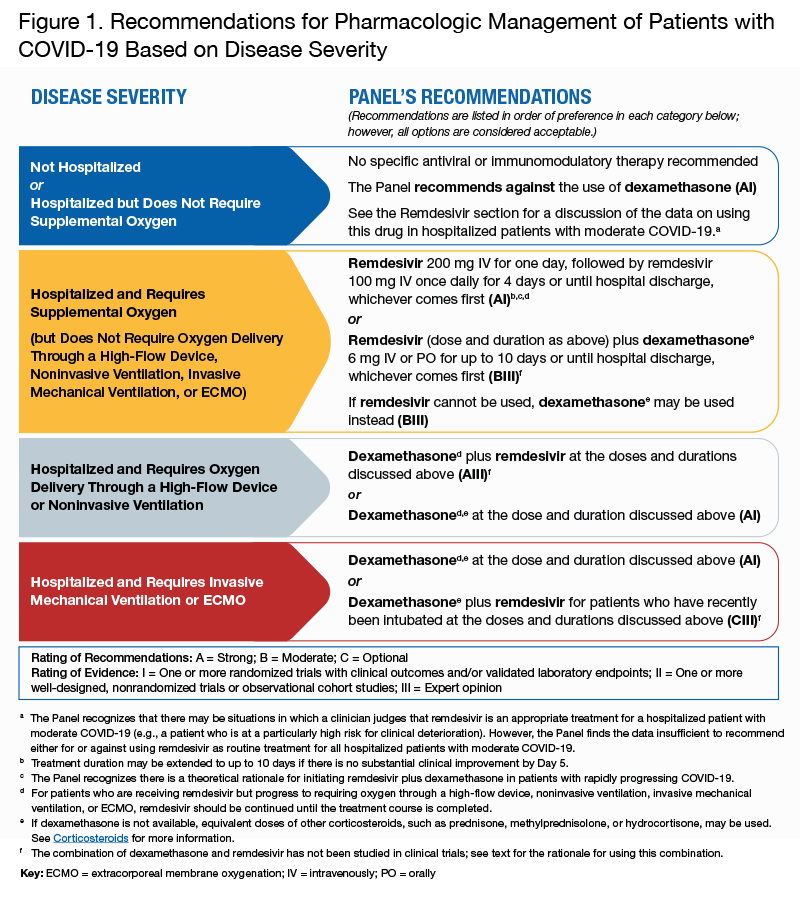
The most recently updated guidelines published October 9, 2020 highlight the Panel’s current recommendations about pharmacologic management of patients with COVID-19. The recommendations are summarized in Figure 1, which is reproduced below. Only two pharmacologic agents, remdesivir and dexamethasone, have been demonstrated to be effective and safe in randomized clinical trials. The benefits of these treatments appears limited to patients with moderate or severe illness. Of note:
- No specific antiviral or immunomodulatory therapy is recommended for patients who are not hospitalized. The panel recommends against using remdesivir for mild illness in outpatient settings.
- There is no role for remdesivir or any anti-viral agent as post-exposure prophylaxis for people who are exposed to SARS-CoV-2 and have no evidence of infection.
- The panel recommends against using dexamethasone in mildly ill patients who do not require supplemental oxygen
- Dexamethasone and remdesivir, either alone or in combination, are recommended for hospitalized patients who require supplemental oxygen, mechanical ventilation, or ECMO.
The panel has separately acknowledged that although convalescent plasma therapy has received Emergency Use Authorization from the FDA for the Treatment of COVID-19, there are currently no data from well-controlled, adequately powered randomized trial that demonstrate the efficacy and safety of this treatment. Based on this, the panel does not recommend either for or against the use of convalescent plasma as a treatment for COVID-19.
A comprehensive summary of clinical data for drugs that are being investigated can be found in the Antiviral Therapy, Immune-Based Therapy, and Adjunctive Therapy sections of the Guidelines.
Panel members include representatives from federal agencies, health care and academic organizations, and professional societies. The names, affiliations, and financial disclosures of the Panel members and ex officio members, as well as members of the support team, are provided in the Panel Roster and Financial Disclosure sections of the Guidelines.
Figure 1. Recommendations for Pharmacologic Management of Patients with COVID-19 Based on Disease Severity
Thank you for your collaboration and support.
Sincerely,
Ryan Westergaard, MD, PhD, MPH
Chief Medical Officer and State Epidemiologist for Communicable Diseases
Wisconsin Department of Health Services
About the COVID-19 Health Alert Network
The content of this message is intended for public health and health care personnel and response partners who have a need to know the information to perform their duties. The HAN will be the primary method for sharing time-sensitive public health information with clinical partners during the COVID-19 response. Health care providers and other officials can subscribe and unsubscribe using their preferred email address at the DHS COVID-19 website.
I received your message
|