|
Recently, we have identified providers who are storing Pfizer COVID vaccine incorrectly. If your clinic orders Pfizer COVID vaccine products, please check to make sure you are storing it correctly according to the COVID Vaccine At-A-Glance Guide.
-
Never store any Pfizer COVID vaccine products in a regular freezer (i.e. where you store other frozen vaccines like Varicella, MMRV, and Moderna COVID vaccine).
- The new Pfizer COVID 12+ pre-filled syringes must be stored in the fridge.
- Single dose vials and multi-dose vials of all Pfizer COVID vaccine products must be stored in an ultracold freezer or the fridge. Vaccine can only be used for up to 10 weeks once placed in the fridge.
- Use Beyond-Use-Date labels:
- When storing single or multi-dose vials in the fridge to indicate the shortened expiration date.
- After puncturing a multi-dose vial to indicate the vaccine can only be used for up to 12 hours after puncture.
- For manufacturer shortened expiration dates
- For reconstituted vaccines.
If you have not yet renewed your provider agreement for the Childhood Vaccine Program in the IIS, please do so now to ensure your continued enrollment in the program. All remaining 2023 provider agreements will expire on April 30th. If you have already submitted your provider agreement for 2024, please disregard this reminder. If you’re not sure if your 2024 agreement is approved, log into the IIS today and check the status. Remember, you will not be able to order childhood vaccines without an approved 2024 agreement.
References:
Penbraya (MenABCWY vaccine) is the first pentavalent meningococcal vaccine licensed for use among persons aged 10-25 years of age to protect against Neisseria meningitidis serogroups A, B, C, W, and Y. Use of the Pfizer Pentavalent Meningococcal Vaccine Among Persons Aged ≥10 Years: Recommendations of the Advisory Committee on Immunization Practices was published on 4/18/24. Here’s a summary of the recommendations:
Penbraya will be available for providers to start ordering through CVP early June. Only providers who chose/use Pfizer brand MenB (Trumenba) will have the ability to order Penbraya due to the inability to interchange MenB vaccine. This was indicated on the vaccine choice form during the selection window that just closed.
Additional resources
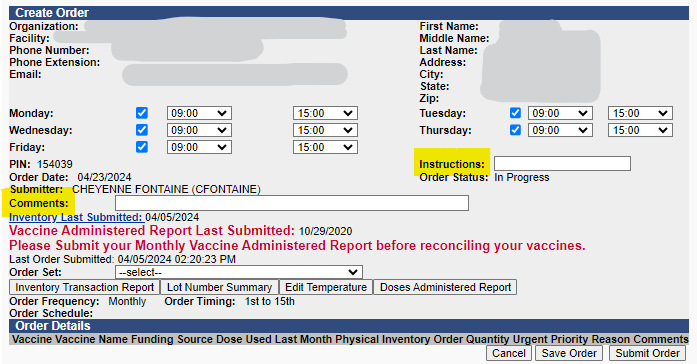
When creating a vaccine order in the IIS, remember to note temporary changes to shipment date/time or other notes to the carrier in the "Instructions" field. These instructions are delivery instructions for the carrier and should only include information relevant to the carrier such as lunch hour closures or directions for which entrance is best for package delivery.
Please note that anything written in the "Comments" section of the order is not exported with your order. This field is for notes to the Childhood Vaccine Program staff reviewing your order.
The Childhood Vaccine Program hosts a monthly series on the third Thursday of every month. We cover a different topic each month and would love to hear what topics you would like to see featured. Email your suggestions to WAChildhoodVaccines@doh.wa.gov.
The CVP Training Series is recorded and posted on the CVP Training webpage. Check out last week's episode on Vaccine Choice and Vaccine Ordering and Receiving!
Next month we will be asking all Childhood Vaccine Program providers to complete our annual Provider Satisfaction Survey. This short survey helps us collect valuable data about your satisfaction with the Childhood Vaccine Program operations and service delivery. Each year, we use this information to improve customer service and provider support.
This year we are excited to host our Provider Satisfaction Survey in RedCAP. Survey instructions will be sent to all vaccine coordinators. We appreciate your help in making Washington's Childhood Vaccine Program the best it can be!
|