|
Providers may notice missing vaccines or no vaccines in the order when trying to receive vaccines.
This is due to a bug in the WAIIS that occurred in orders submitted between January 31st and February 6th where the NDCs dropped from random vaccines in random orders.
If there are other vaccines that show in the order receive these through the order and this should remove the order from the Inbound Order list.
For vaccines that are not showing in the inbound order, those will have to be manually added through the Search/Add feature under Lot Numbers. See the inventory guide for instructions (pgs. 1-2).
We are sorry for the inconvenience this is causing. We are working to resolve the bug and find a way to clear those blank orders out of the Inbound Orders.
Please join us this Thursday, February 15th, 2024 at noon to learn how to renew your Childhood Vaccine Program provider agreement in the IIS.
Topic: Provider Agreement Renewal Process
Date: February 15, 2024
Time: 12 noon
Intended Audience: Providers and Vaccine Coordinators for the Childhood Vaccine Program
Join the Training Session
Webinar ID: 851 4240 2545
Passcode: 673609
It is almost that time of year again! Childhood Vaccine Program provider agreement renewal will begin in the IIS on March 1st. Renewal is organized by county so be sure to check the Renewal Schedule to find yours. There are many resources available to help you with this process:
You can get a head start on renewal by completing the following:
- check all thermometer calibrations are up to date.
- gather your practice profile data for 2023 which includes the number of children served in your clinic by age range and eligibility status.
- primary and back-up vaccine coordinators should complete the required You Call the Shots training modules listed below.
Prepare now for provider agreement renewal by completing the two You Call the Shots courses that are required annually for all primary and back-up vaccine coordinators.
Follow these instructions to complete the courses, then take the post-test and course evaluation through the CDC TRAIN site. After successfully completing these steps, you will receive a training certificate. These training certificates should be kept with all other program documentation in your Childhood Vaccine Program binder or files.
When searching the course catalog in TRAIN, please use the course numbers shown above to ensure you complete the correct courses. Please do not complete the You Call the Shots AUDIT ONLY VERSION trainings like the one shown below.

FluMist vaccine is no longer available to order. Many providers had submitted orders for FluMist in mid-January before we knew that this vaccine would no longer ship out.
The orders of FluMist placed since mid-January will not ship. McKesson stopped shipping FluMist at that time due to the short expiration date in relation to the order date.
If you have pending FluMist vaccine to receive in the IIS, please reject that vaccine in your Inbound Order page in the IIS to clear that order out.
We apologize for the inconvenience.
Seasonal administration of Abrysvo for pregnant people ended January 31st, 2024 in Washington state. Abrysvo will not be available for ordering for pregnant teens from the Washington Childhood Vaccine Program as of February 1st.
Remaining Abrysvo in your inventory should be held until its expiration date. Abrysvo shipped through the CVP does not expire until March 2025 and can be used for the next season.
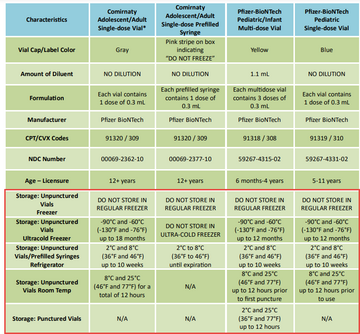
Recently, we have identified providers who are storing Pfizer COVID vaccine incorrectly. If your clinic orders Pfizer COVID vaccine products, please check to make sure you are storing it correctly according to the COVID Vaccine At-A-Glance Guide.
-
Never store any Pfizer COVID vaccine products in a regular freezer (i.e. where you store other frozen vaccines like Varicella, MMRV, and Moderna COVID vaccine).
- The new Pfizer COVID 12+ pre-filled syringes must be stored in the fridge.
- Single dose vials and multi-dose vials of all Pfizer COVID vaccine products must be stored in an ultracold freezer or the fridge. Vaccine can only be used for up to 10 weeks once placed in the fridge.
- Use Beyond-Use-Date labels:
- When storing single or multi-dose vials in the fridge to indicate the shortened expiration date.
- After puncturing a multi-dose vial to indicate the vaccine can only be used for up to 12 hours after puncture.
- See additional guidance below for BUDs when storing single or multi-dose vials at room temperature.
|