 A heartfelt thank you for all of your efforts to participate in the Adult Vaccine Program to serve uninsured Washingtonians! Your dedication plays a crucial role in ensuring that all adults have the opportunity to be vaccinated. We are so appreciative of your partnership and looking forward to continuing to work together in 2024!
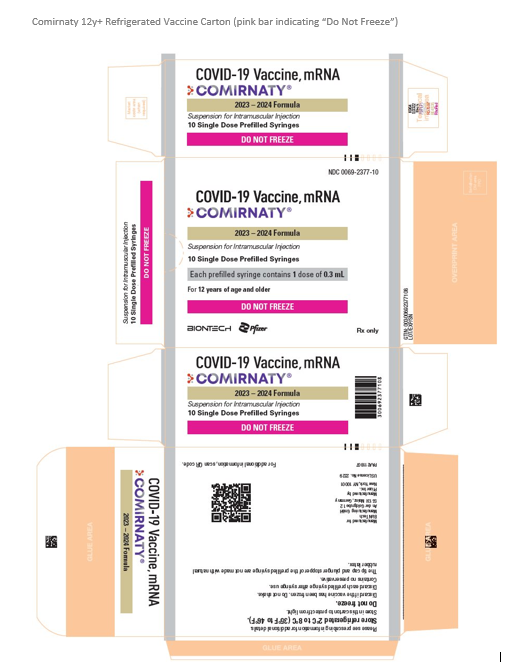
 The current Pfizer COVID-19 vaccine for persons 12y+ (NDC 00069-2362-10) on CDC’s pediatric and adult contracts has been replaced with a new presentation. This new presentation is a carton of 10 prefilled syringes (NDC 00069-2377-10). It is a refrigerated formulation that has never been frozen, must be stored at 2-8C, and whose shelf life aligns with the expiration date printed on the carton and syringe labels.
Some important information to note:
- This new presentation MUST BE STORED BETWEEN 2°C and 8°C (36°F and 46°F). Do NOT store at ultra-cold or standard freezer temperatures.
- These pre-filled syringes can be used through the expiration date printed on the carton. The 10-week beyond-use date (BUD) for refrigerator storage does not apply.
- This guidance applies to this presentation ONLY. Pfizer-BioNTech COVID-19 vaccines for children aged 6m-4y and 5-11y, which are shipped to providers in CDC’s programs directly from Pfizer on dry ice and can be stored at either at ultracold temperatures until expiry or up to 10 weeks at 2-8C.
Of note: Pfizer will continue to allow providers to privately order the original single dose vial (SDV) presentation for age 12+. Therefore, providers may have BOTH presentations – SDV with a 10-week BUD and pre-filled syringe that can be used through the expiration date - in their storage units. If this occurs, please educate staff on the different storage requirements and strategies to prevent potential errors. Clinical materials are being updated to include this new presentation and storage guidance.
Below, you will find an image of the carton for this vaccine. The carton includes a bright pink stripe that includes the words “DO NOT FREEZE” to alert the provider about the storage and handling requirements for this vaccine.
 Providers who have the original Pfizer 12y+ vaccine on hand can and should continue to use this product until it is consumed, expired, or has been stored at 2-8C for longer than the allowable 10 weeks.
Our 2023-2024 State Supplied COVID-19 Vaccines-At-a-Glance Guide has been updated for your reference.
 CDC has received several reports of vaccine providers who have inadvertently administered RSV vaccines (Arexvy or Abrysvo) to infants, rather than giving them the RSV monoclonal antibody product recommended for infants. The recommended immunizing product (RSV monoclonal antibody) for infants is nirsevimab (trade name: Beyfortus, commercialized by Sanofi and manufactured AstraZeneca).
As a reminder, RSV vaccines (Arexvy or Abrysvo) are not approved or recommended for use in infants and young children. These RSV vaccines have not been studied in infants and young children; therefore, vaccine safety and effectiveness for this population are unknown.
Vaccine providers who carry both nirsevimab for use in infants and young children and RSV vaccines for use in adults and pregnant people should be especially diligent in following vaccine administration safety procedures to prevent errors.
For more information, please refer to the following CDC resources:
Vaccine Administration
We encourage providers to promptly report vaccine administration errors to the Vaccine Adverse Event Reporting System (VAERS). In the event that a vaccine administration error occurs, please reach out to CDC at NIPINFO@cdc.gov for further guidance.
 With holidays quickly approaching, please be mindful of shipping delays, limited or no shipping days, and plan your adult COVID-19 and flu vaccine orders accordingly. Please ensure your accountability reports are up to date prior to placing orders to ensure expedited processing. It's also a good idea to update your shipping hours in your provider agreement, if needed. Check out the holiday shipping calendar on our website for more information.
|