|
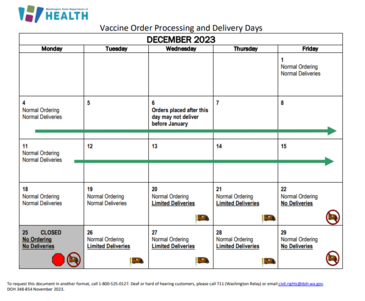
With the holidays coming up very quickly, please place your vaccine orders in the IIS by December 6th, 2023 if you need vaccine prior to the new year. Holiday shipping is limited, so planning ahead to avoid running out of vaccine is critical. Please place orders by Wednesday and ensure your accountability reports are up to date to ensure a speedy approval process. You can refer to the holiday shipping calendar here.
 Vaccine Ordering Reminders:
- Ordering COVID-19 vaccine is required for all enrolled providers because it is an ACIP recommended vaccine, unless your clinic is designated as a "specialty provider" (ie: birthing hospital etc).
- Td and PPSV23 can be ordered in single doses to avoid waste.
- The RSV product, nirsevimab, is currently under allocation and only available for specific providers.
- Orders can be placed monthly to allow for more flexibility.
- Please plan to always have a 30 day safety stock on hand to avoid running out of vaccine.
As you may have heard, the Childhood Vaccine Program has recently launched the REDCap portal for temperature log submissions! Providers enrolled in the Adult Vaccine Program (AVP) only and those dual enrolled in both the Childhood Vaccine Program (CVP) and Adult Vaccine Program are required to submit temperature logs through their REDCap portal beginning December 1st. Every primary and back up vaccine coordinator should have received an email with the link to access your clinic's portal on December 1st.
All other providers can choose the option to upload temperature logs into REDCap this month, however, it is not required for providers only enrolled in CVP until January 1, 2024. If you were unable to attend our recent CVP Training Series on this new process, please view the recording for Navigating Temperature Log Submission in REDCap. Still have questions? Drop into the Office Hours and ask!
Office Hours: REDCap Provider Portal for AVP and CVP Providers
This is an opportunity for providers to drop-in and ask questions about the new REDCap Provider Portal.
Dates
- Dec 6, 2023
- Dec 13, 2023
- Dec 20, 2023
- Dec 27, 2023
- Jan 3, 2024
- Jan 10, 2024
- Jan 17, 2024
Time: 12:00-1:00 PM
Intended Audience: Providers and Vaccine Coordinators for the Childhood Vaccine Program and Adult Vaccine Program
Join the Zoom Meeting
Meeting ID: 816 0499 1181
Passcode: 865423
Join us for the next session in the CVP Training Series on December 14, 2023 at 12:00 pm for a timely training about what to do during a power outage and how to handle reporting temperature excursions in the new REDCap portal.
CVP Training Series: What to Do In a Power Outage, Preparing Your Emergency Transport Plan and Reporting Temperature Excursions in REDCap
Date: December 14, 2023
Time: 12 noon
Intended Audience: Providers and Vaccine Coordinators for the Childhood Vaccine Program
Join the Training Session
Webinar ID: 851 4240 2545
Passcode: 673609
For School Based Health Clinics that will be closing for the upcoming winter break, we want to remind you of the Childhood Vaccine Program’s (CVP) Facility Closure Policy which is located on page 19 of the Vaccine Management Plan:
- If your clinic will be closed for 10 days or less, you will need to have someone go in and check on the vaccines every five days at a minimum (min/max and current temps).
- If your clinic will be closed for more than 10 days, you will need to have someone go in and check on the vaccines at least every five days.
- If this isn’t possible (i.e., you don’t have access to the building), you will need to transport the vaccines to another CVP provider prior to school closure. The vaccines should continue to be monitored in the back-up location with the 2x/day temperature checks being recorded on the paper temperature log. If you need to transport your vaccine to a back-up facility, please be sure to follow the Vaccine Transport Guidelines.
- Once facility operation resumes, vaccines can be transported back to the facility of origin. Review temperature data to ensure the storage unit is working properly before transporting and placing vaccine back inside the unit.
|
On 08/21/2023, the FDA approved sterile diluent supplied in a prefilled syringe for vaccines in Merck’s MMRV Family. These include M-M-R®II (Measles, Mumps, and Rubella Virus Vaccine Live), VARIVAX® (Varicella Virus Vaccine Live), and ProQuad® (Measles, Mumps, Rubella and Varicella Virus Vaccine Live). When reconstituting these vaccines using the prefilled diluent syringe, the need to withdraw the sterile diluent from a vial is eliminated.
|
|
Please note that this update only impacts the sterile diluent, and there is no change to the vials containing lyophilized M-M-R®II, ProQuad, or VARIVAX (ie, packaging, NDC, etc.).
|
|
•
|
Early in the 4th quarter of 2023, the transition from vial diluent to a prefilled diluent syringe began. During the transition period, you may receive either the vials or syringes of diluent with your order of M-M-R®II, ProQuad, or VARIVAX.
|
|
|
•
|
The exact timing of the transition will vary based on the vaccine and the ordering channel (ie, direct from Merck, wholesaler, distributer, or CDC).
|
|
|
•
|
The transition to the prefilled diluent syringes should be fully complete by the end of 2023.
|
|
|
•
|
The vaccines and sterile diluent will continue to be packaged separately, allowing you to store the diluent separately from the lyophilized vaccines. Store the prefilled diluent syringes at room temperature (68°F to 77°F, 20°C to 25°C) or in the refrigerator (36°F to 46°F, 2°C to 8°C). Do not freeze the sterile diluent.
|
|
|
•
|
The sterile diluent will be supplied as a carton of 10 single-dose prefilled diluent syringes (NDC: 0006-4175-88). The carton dimensions are: 4.29” x 5.236” x 1.397”.
|
|
|
•
|
When shipped directly from Merck, orders of 40 doses (4 cartons) or fewer of M-M-R®II, ProQuad, and VARIVAX will be shipped together with the prefilled diluent syringes in one shipment.
|
|
|
•
|
When shipped directly from Merck, orders of 50 doses (5 cartons) or more of M-M-R®II, ProQuad, and VARIVAX will be shipped in two parcels. The diluent will be shipped in an ambient temperature parcel.
|
|
| |
| |
|