|
With the vote to include Nirsevimab in the Childhood Vaccine Program, we are excited to announce that we have added this product to all routine childhood vaccine order sets in the IIS. Nirsevimab is a monoclonal antibody product used to prevent serious respiratory syncytial virus (RSV) illness in babies. This means enrolled health care providers are expected to have the product available for all babies who are at risk for severe RSV illness. Due to the cost of this product please order only as needed. It will ship with all other routine vaccines and should arrive in provider offices one to two weeks after placing an order.
Nirsevimab is the new monoclonal antibody product recommended for infants younger than 8 months of age entering their first RSV season if:
- The mother did not receive RSV vaccine (ABRYSVO) during pregnancy
- The mother’s RSV vaccination is unknown
- The infant was born within 14 days of maternal RSV vaccination
ABRYSVO, an RSV vaccine, was recently recommended for pregnant persons during 32 through 36 weeks by the Advisory Committee on Immunization Practices.
The following children 8 through 19 months old who are at increased risk for severe RSV disease and entering their second RSV season are recommended to get nirsevimab:
- American Indian or Alaska Native children
- Children with chronic lung disease of prematurity who require medical support during the six months before the start of their second RSV season
- Children with severe immunocompromise
- Children with cystic fibrosis
Give nirsevimab intramuscularly in the anterolateral thigh. Here are dosing details:
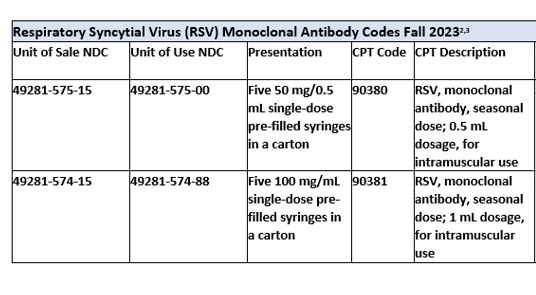
- Infants born during the RSV season should receive a single dose of nirsevimab (50 mg for infants <5 kg and 100 mg for infants ≥5 kg) in the first week of life. The dose can be administered either in the hospital or outpatient clinic. Infants with prolonged hospitalization (e.g., preterm infants) should be immunized shortly before or after discharge.
- In infants younger than age 8 months born outside the RSV season, a single dose of nirsevimab (50 mg for infants <5 kg and 100 mg for infants ≥5 kg) is recommended. Administration should be continued during the RSV season for those who have not received a dose.
- In children ages 8 through 19 months who are at increased risk of severe RSV disease, administer a single 200 mg dose of nirsevimab during the season for those who have not received a dose.
You can find more details on this exciting product at the following link: Healthcare Providers: RSV Immunization for Children 19 Months and Younger | CDC
The storage of the nirsevimab product is the same as with routine vaccines. It should be stored in the refrigerator between 36°F to 46°F (2°C to 8°C). After removal from the refrigerator, this product must be used within 8 hours. Please store this product in its original carton to protect from light until time to use. Do not freeze, shake, or expose to heat.
PLEASE NOTE that until July 1, 2024, you should NOT bill commercial carriers and third-party administrators (TPA) a dosage-based assessment (DBA) for the nirsevimab vaccine cost. DBAs are NOT required for vaccines until they are published on the WVA Vaccine Assessment Grid.
• Your practice should work directly with the patient's insurance carrier or TPA for reimbursement of the administration fee.
• Your practice will still be required to use the V25 Eligibility Code during the WA DOH eligibility screening process.
More information will follow in the spring of 2024 regarding the addition of nirsevimab to the WVA Vaccine Assessment Grid. Please read the WVA Provider Bulletin for more information.
Department of Health RSV Webpage
CDC's RSV Landing Page
Healthcare Providers: RSV Immunization for Children 19 Months and Younger | CDC
|