|
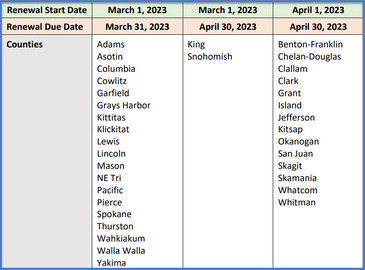
Some providers enrolled in the Childhood Vaccine Program are required to renew their provider agreement by the end of March. Please check out the Renewal Schedule to see if your clinic is located in one of these counties. If you are due to renew your provider agreement in March, and it isn't complete by this Friday, March 31st, 2023, your clinic will no longer be allowed to order childhood vaccines until your 2023 agreement has been approved. Please submit your 2023 provider agreement today to avoid an interruption in your vaccine ordering privileges.
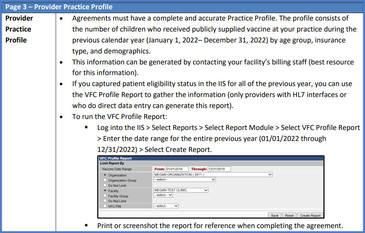
Provider Support Specialists with the Childhood Vaccine Program are busy reviewing Provider Agreement renewals for 2023 and noticing that the practice profile page in the agreement may be causing confusion. On page 3 in the provider agreement, you are required to enter the number of children who received publicly supplied vaccines in your clinic in the previous calendar year by age group and eligibility/insurance status. This data can be collected in a variety of ways, either working with your billing department to generate this data, or by running the VFC Profile Report in the IIS. Keep in mind that when you are renewing your agreement, the previous year's data will auto-populate the page, so you need to update it with 2022 data. If it's not updated, we will return the agreement to you for updates. Please ensure that when you submit your provider agreement for approval, that your practice profile data has been updated with 2022 data. Use the Provider Agreement Renewal Guide to assist you with the renewal process.
Join us Thursday, April 20, 2023 for the next session in the monthly CVP Training Series. This session will give an overview of vaccine choice and review the vaccine ordering process. Vaccine choice is the window of time twice per year when a provider has the opportunity to switch their preferred vaccine brands in their order set. Our Immunization Nurse Consultant will also cover new vaccines recently approved, considerations for using these vaccines in your practice, and share available clinical resources. Please join us to learn more! You can also find previously recorded sessions in the CVP Training Series on our webpage.

 |
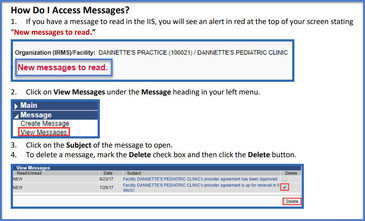
Did you know you can see messages in the IIS that your Provider Support Specialist has sent you? Often times these messages are followed up with a typical email, but you can also see messages directly in the IIS if your Provider Support Specialist is trying to reach you. For example, you may see messages requesting additional accountability reports that may be missing in order to approve a vaccine order or vaccine transfer. You may also see messages if your provider agreement is returned for updates or is approved or expired. To view these messages, use this guide.
|