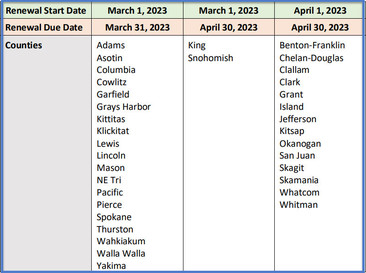
 The 2023 Provider Agreement renewal period begins March 1st, depending on what county you are located in. Please review the renewal schedule above to learn when your clinic should begin the renewal process in the IIS. In order to remain enrolled in the Childhood Vaccine Program, a renewed provider agreement is required to be submitted in the IIS by your due date. We have many resources available to assist you including an upcoming CVP training this Thursday.
Resources:
Please join us this Thursday for a very important training session that will cover how to renew your Childhood Vaccine Program provider agreement in the IIS. Renewals begin March 1st for some counties. We look forward to another great session of the CVP Training Series.
Date: February 16, 2023
Time: 12:00 (noon)
Topic: Provider Agreement Renewal Process
Intended Audience: Providers and Vaccine Coordinators
The You Call the Shots training modules have been updated for the new year. Please ensure your primary and back up vaccine coordinators complete both modules before you renew your provider agreement. These training dates are required in your 2023 provider agreement.
You Call the Shots Trainings:
- Vaccines For Children (VFC)
- Vaccine Storage and Handling
In addition, please ensure all vaccine thermometers have current certificates of calibration, including your back up thermometer. The calibration dates are required when renewing your provider agreement in the IIS.
The 2023 Immunization Schedules are now available. Main changes to the schedules include:
- COVID-19 vaccines were added for both children and adults in the Tables and Notes sections of the Schedules.
- Addition of PCV15 vaccine recommendations to the child and adolescent schedule.
- Addition of the new MMR (Priorix) and Hepatitis B (PreHevbrio) vaccines to the child and adult schedule.
- Recommendation for an additional third dose of MMR vaccine for persons at increased risk during a mumps outbreak
- Language added that the newly licensed Menveo one-vial (all liquid) formulation should not be administered before age 10 years.
- For persons aged 18 years and older at increased risk of exposure to poliovirus, the recommendation is to complete a 3-dose series or for one lifetime IPV booster if a person has evidence of a completed polio vaccination series.
- PCV15 and PCV20 vaccine recommendations were added to the adult schedule.
- The zoster vaccination note was updated to provide guidance for patients with serologic evidence of prior varicella susceptibility, and guidance for persons with immunocompromising conditions who do not have a documented history of prior varicella infection, varicella vaccination, or prior herpes zoster.
Correction to image previously shared in Vaccine Blurbs Issue 159 on November 22, 2022.
No mixing, no diluting: FDA licenses new Rotarix liquid formulation
On November 4, FDA licensed a new formulation of Rotavirus Vaccine, Live, Oral (Rotarix, GSK). The new formulation is free of porcine circovirus and comes as a liquid that does not require mixing or dilution. Both the lyophilized (requires reconstitution) and liquid rotavirus vaccine presentations will remain in use until the lyophilized formulation is used up or expires.
 Related Links
|