|
If you receive flu vaccine through the Childhood Vaccine Program, print out or bookmark a great reference showing the flu vaccines and recommended age/dosages.
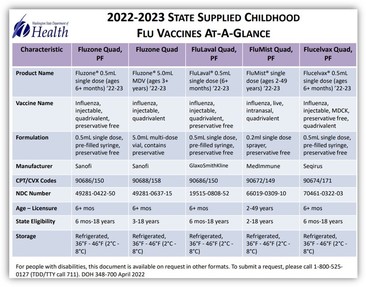 According to Washington state law (RCW 70.95M.115), children under 3 years of age and pregnant persons should NOT be given vaccines that contain more than trace amounts of mercury (thimerosal).
Use the following flu vaccines for the following groups (follow appropriate age guidance for each vaccine):

Please give a full 0.5mL dose for all children 6 months and older if using Fluzone, FluLaval, or Flucelvax. There used to be a 0.25mL dose in the past for children 6 months-35 months, but this formulation is no longer available. If the child needs a second dose during the flu season (please see article below), then give another full 0.5mL dose at least 4 weeks later. Please do NOT give half of the 0.5mL dose for younger children. The Immunize.org’s Ask the Experts, flu section provides a comprehensive Q&A, including steps to take if a vaccine administration error occurred.
Children aged 6 months through 8 years require 2 doses of influenza vaccine administered a minimum of 4 weeks apart during their first season of vaccination if:
- They are receiving flu vaccine for the first time
- They did not receive a total of at least two doses of flu vaccine before July 1, 2022 or
- Vaccination history is unknown
Patients who received two or more doses at any time before July 1, 2022 only need one dose. Review the Guide for Determining the Number of Doses of Influenza Vaccine to Give to Children Age 6 Months Through 8 Years for more details.

Did you miss the August session?
If you missed August's CVP training session that covered back to school prep, the new clinic move policy, and off-site clinic guidelines, you can access it now on our training webpage.
Take a look at all our recorded training sessions!
September's CVP training session will cover 2022-23 flu season information so make sure to join.
September 15, 2022
12:00 pm
CVP Training Series: 2022-2023 Flu Vaccine Update
Intended Audience: Providers and Vaccine Coordinators
|