|
March 17, 2022 12:00-12:30PM
CVP Training Series: WVA Billing & Eligibility
Visit our webpage to view previously recorded training sessions, get additional training resources, and to see what topics are coming up next:
April 21, 2022 – CVP Training Series: Ordering Basics
Starting April 1, the Childhood Vaccine Program (CVP) will begin accepting monthly temperature summary reports from Digital Data Loggers (DDL) and Temperature Monitoring Systems (TMS), upon program approval. Providers wishing to obtain approval must submit a copy of their report to the program at WAChildhoodVaccines@doh.wa.gov for review.
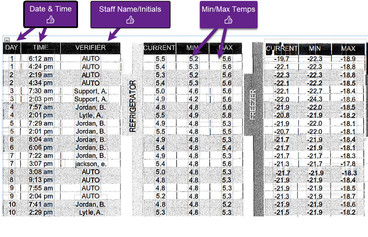
To be considered for approval, DDL and TMS summary reports must contain the following as required by CDC’s Vaccines for Children Program:
-
Min/max temperature check once per day
-
Time and date of reading
-
Name or initials of staff member who recorded the reading
Sample TMS that meets criteria:
Why are we making this change?
On April 1, 2022 the COVID Vaccine Program will begin requiring providers to submit monthly temperature logs in either handwritten paper form or DDL/TMS summary reports. While this is a new requirement for COVID vaccine providers, the Childhood Vaccine Program has had a temperature reporting requirement for many years. We are making this change to align with COVID vaccine reporting options, while still meeting the CDC’s requirements for the Vaccine for Children program. Providers whose DDL or TMS reports do not meet the reporting requirements listed above must continue to submit monthly paper temperature logs.
Monitoring vaccine storage equipment and temperatures are daily responsibilities to ensure the viability of your vaccine supply and the safety of your patients. Implementing routine monitoring activities can help you identify temperature excursions quickly and take immediate action to correct them, preventing loss of vaccines and the potential need for revaccination of patients.
Childhood and adolescent immunization rates have remained low compared to pre-pandemic rates even after the stay at home order was lifted. The Office of Immunization created a survey to get feedback from immunization providers on barriers to routine pediatric and adolescent vaccines. It should take approximately 5 minutes to complete and would help us to understand factors that might be contributing. Please complete the survey by Friday, March 25th.
Here is the link to the survey.
For any questions or feedback, please email immunizewa@doh.wa.gov.
The IACW quarterly meeting will be held on April 27, 2022, the same time as National Infant Immunization Week. National Infant Immunization Week (NIIW) is a yearly observance highlighting the importance of protecting children two years and younger from vaccine-preventable diseases (VPDs). This year, in particular, it’s critical to ensure that families stay on track for children’s routine checkups and recommended vaccinations — even during COVID-19.
We are honored to have UW Medicine present a new campaign “One Vax Two Lives” to dispel COVID-19 vaccine misinformation for pregnant and breastfeeding individuals. This group continues to make inroads into vaccine hesitancy related to women's health issues including menstruation, fertility and safety in pregnancy and breastfeeding. Also, there is a panel to discuss challenges in vaccinating infant and pregnant women, promoting COVID-19 vaccines among pregnant women, and vaccine access/barriers experienced among this population and resources.
Highlight:
• When Vaccine Misinformation Targets Women’s Reproductive Health and Pregnancy: Fighting Back with the One Vax Two Lives Campaign
• Panel on increasing awareness about vaccinations for Pregnancy and infants
 Immunize.org’s laminated schedules for children and adults are available for order. These laminated schedules are great for any busy healthcare setting where vaccinations are given since they can be wiped down and durable enough to stand up to a year's worth of use. The laminated schedules are now published in a standard 8.5” X 11” booklet format. The child/adolescent schedule is a total of five double-sided pages and includes a 1-page handout summarizing the dose, route, and needle size recommendations.
|