|
|
This weekly newsletter provides summarized content on COVID-19 vaccine related topics. This broad level of information is tailored towards health care professionals, partners, and the general public.
WASHINGTON COVID-19 VACCINATION STATUS: ALL AGES 5 AND UP
|
|
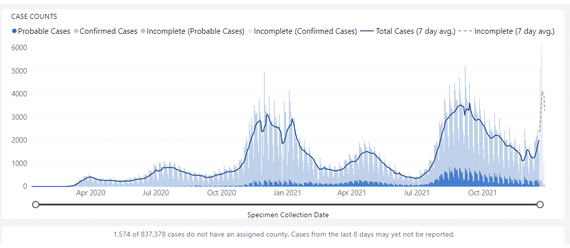 COVID-19 CASES SPIKE SIGNIFICANTLY IN THE PAST WEEK
The state of Washington is seeing a spike in COVID-19 cases, and transmission is expected to rise in the coming weeks. While it is still too early to tell how much of the increase is due to Omicron, epidemiologists with the Washington State Department of Health (DOH) agree that Omicron prevalence is increasing and is most likely the dominant strain.
While additional data are still being sent to the state, the largest single-day report of new cases so far occurred on December 24, with 6,140 new COVID-19 cases. Despite a recent increase in testing around the holidays, public health officials say the increase in new cases significantly outpaces the increase in testing. The number of cases is expected to continue to increase through the new year.
The spike in cases, together with the first cases of the flu this season, will likely mean increased hospitalizations in the near future, raising concerns about the state’s health care system. Washington state’s hospitals and clinics are already stretched and strained due to an exhausted and understaffed workforce who have been caring for more patients than ever before.
Read the full news release here.
|
|
WHAT TO DO IF YOU TEST POSITIVE FOR COVID-19
The Washington State Department of Health (DOH) is offering a detailed reminder about what people should do if they test positive for COVID-19. Cases are rising sharply, driven by the omicron variant, which is believed to be more contagious than previous variants, including delta. Tracking cases and preventing further spread of disease starts with testing. Anyone with any signs or symptoms of COVID-19 or known exposures should get tested, regardless of vaccination status or prior infection.
Please read the full news release to see the checklist.
|
|
DEPARTMENT OF HEALTH UPDATES ISOLATION AND QUARANTINE GUIDANCE TO ALIGN WITH CDC RECOMMENDATION
EL DEPARTAMENTO DE SALUD ACTUALIZA LAS ORIENTACIONES SOBRE EL AISLAMIENTO Y LA CUARENTENA PARA ADAPTARLAS A LA RECOMENDACIÓN DE LOS CDC
|
|
|
Today, the Washington State Department of Health (DOH) announced updates to state isolation and quarantine guidance, in alignment with the newly released recommendations from the Centers for Disease Control and Prevention (CDC).
Monday, the CDC announced the following shift in isolation and quarantine recommendations for the general public:
-
Shorten the recommended time for isolation from 10 days for people with COVID-19 to five days, if they are asymptomatic on day five, followed by five days of wearing a mask when around others. If symptoms occur, individuals should immediately quarantine until a negative test confirms symptoms are not related to COVID-19.
- Recommended quarantine period for those exposed to COVID-19
- For unvaccinated individuals or those that are more than six months from their original mRNA dose or more than two months after the J&J vaccine and not yet boosted, CDC is recommending quarantine for five days followed by strict mask use for another five days.
- Vaccinated individuals who have received their booster do not need to quarantine but should wear a mask for 10 days after exposure.
- If symptoms do occur after exposure, individuals should immediately quarantine until a negative test confirms symptoms are not related to COVID-19.
- When in doubt, get tested on day five and as always, wear a mask when in public spaces after isolation and quarantine are complete. Also be mindful of activities, especially indoors and in crowded settings.
Read the full news release in English, which includes updated healthcare personnel guidance.
|
|
Hoy, el Departamento de Salud del Estado de Washington (DOH, por su sigla en inglés) anunció actualizaciones de las orientaciones estatales sobre el aislamiento y la cuarentena, en consonancia con las nuevas recomendaciones de los Centros para el Control y la Prevención de Enfermedades (CDC, por su sigla en inglés) (solo en inglés).
El lunes, los CDC anunciaron los siguientes cambios en las recomendaciones de aislamiento y cuarentena para el público en general:
-
Acortar el tiempo de aislamiento recomendado de 10 días para las personas con COVID-19 a cinco días, si son asintomáticos en el quinto día, seguido de cinco días de uso de una mascarilla cuando estén cerca de otras personas. Si presentan síntomas, las personas deben ponerse inmediatamente en cuarentena hasta que obtengan un resultado negativo en una prueba que confirme que los síntomas no están relacionados con la COVID-19.
- Período de cuarentena recomendado para los expuestos a la COVID-19
- Para las personas no vacunadas o que hayan pasado más de seis meses desde su dosis original de ARNm o más de dos meses después de la vacuna de J&J y que aún no hayan recibido una vacuna de refuerzo, los CDC recomiendan la cuarentena durante cinco días, seguida del uso estricto de mascarilla durante otros cinco días.
- Las personas vacunadas que recibieron su vacuna de refuerzo no necesitan hacer cuarentena, pero deben usar mascarilla durante 10 días después de la exposición.
- Si presentan síntomas después de la exposición, las personas deben ponerse inmediatamente en cuarentena hasta que obtengan un resultado negativo en una prueba que confirme que los síntomas no están relacionados con la COVID-19.
- En caso de duda, hágase la prueba el quinto día y, como siempre, use mascarilla cuando esté en espacios públicos después de completar el aislamiento o la cuarentena. También hay que estar atento a las actividades, sobre todo en interiores y en lugares con mucha gente.
Leer el artículo en español.
|
|
CDC UPDATES AND SHORTENS ISOLATION AND QUARANTINE PERIOD FOR GENERAL POPULATION
On Monday, December 27, 2021, the Centers for Disease Control and Prevention put out a news release that shortens the recommended time of isolation from 10 days for people with COVID-19 to 5 days, if the person does not have symptoms. People should wear a mask around others for 5 days after isolation ends.
In addition, the CDC is updating the quarantine period for those exposed to COVID-19. People who are unvaccinated or six months past completing their COVID-19 vaccine series should quarantine for 5 days and wear a mask around others for an additional 5 days. If quarantine is not feasible, the person should wear a mask at all times around others for 10 days after exposure.
Individuals who received a booster shot do not need to quarantine following an exposure, but should wear a mask for 10 days after exposure.
Read the full news release on the CDC website.
|
|
AT-HOME COVID-19 TESTS - WHY THEY SHOULD BE ON YOUR SHOPPING LIST
Planning to get together with family and friends? Whether you’re traveling or hosting, you should consider adding at-home COVID-19 tests to your checklist.
At-home tests are a simple way to provide a little peace of mind for your gathering. They’re quick, painless, portable, and priced at $25 on average– making them an easy option for a variety of occasions.
Here are some of the basics you should know about at-home testing.
An at-home test is one of several options for testing for the COVID-19 virus. Like the name suggests, you can do this test at home — or anywhere. This kind of test is also known as a self-test or an over-the-counter (OTC) test.
You do the test on yourself and read your own results. You can also help your kids or other family members take an at-home test.
There are nearly a dozen over-the-counter tests authorized by the FDA for emergency use. Most at-home tests are rapid (antigen) tests. These rapid tests provide results within 15–30 minutes. There are also PCR at-home tests, which require you to send your sample to a lab; this takes at least a day to get results. These are technically known as “at-home collection tests.”
Read more on the DOH blog page.
|
|
 RECOMENDACIONES PARA VIAJAR Y DISFRUTAR LA TEMPORADA DE FIESTAS Y FIN DE AÑO
¿Se está preparando para viajar durante las vacaciones? Si es así, usted no está solo. Los expertos predicen que los viajes durante la temporada de fiestas podrían aumentar hasta un 80% (solo en inglés) comparado con el año pasado.
La realidad es que viajar durante las vacaciones y temporada de fiestas puede ser estresante, sin importar las condiciones. La propagación del COVID-19 y sus variantes añade un elemento adicional de incertidumbre e inseguridad.
La buena noticia es que si usted está completamente vacunado, su viaje debería ser seguro, siempre y cuando siga las recomendaciones adecuadas durante el viaje. A continuación, le haremos saber cómo puede mantenerse a usted mismo y las personas que lo rodean a salvo mientras viaja por avión y por las carreteras en esta temporada de fiestas.
Leer el artículo en español.
|
|
 TEST POSITIVE AT HOME? HELP STOP THE SPREAD WITH WA NOTIFY AND AT-HOME COVID TESTING
You weren’t feeling well and took an at-home, over-the-counter COVID-19 test and it’s positive. What’s next? In addition to isolating and informing close contacts, you can now easily use your smartphone to help stop the spread of COVID-19. If you have a smartphone with WA Notify enabled, you can request a verification code to alert other WA Notify users who may have been exposed.
What is WA Notify?
WA Notify is a free exposure notification tool that works on smartphones to alert users if they may have been exposed to COVID-19. It’s completely private; no personal information is shared, including where or exactly when the exposure happened. WA Notify was launched a year ago and since then, over 2.75 million people have enabled the tool on their phones.
The app runs in the background on smartphones that have enabled WA Notify; the phones will exchange random, anonymous codes when near others who have also enabled WA Notify on their phones. If a user later tests positive for COVID-19, and adds a verification code into the app, other WA Notify users they were near will receive an anonymous notification that they may have been exposed. These close contacts can then take steps to help stop the spread of COVID-19, like testing and quarantining.
Read the full DOH blog.
|
|
FDA APPROVES SHELF-LIFE FOR PFIZER COVID-19 VACCINE
The Food and Drug Administration (FDA) has approved a shelf-life extension for the Pfizer pediatric vaccine (orange cap) and the adult Tris-Sucrose vaccine (gray cap). The expiration date is now 9 months from manufacture date. These extensions are effective immediately and only apply to frozen ultra low temperature (ULT) inventories.
The updated expiry dates for both the orange and gray cap vials have been extended to 9 months from the manufacturer date while stored at frozen ULT. Please see the chart below for vaccine date extensions.
|
Printed Manufacturing Date
|
9-Month Expiry Date*
|
|
06/2021
|
Feb. 28, 2022
|
|
07/2021
|
Mar. 31, 2022
|
|
08/2021
|
Apr. 30, 2022
|
|
09/2021
|
May 31, 2022
|
|
10/2021
|
Jun. 30, 2022
|
|
11/2021
|
July. 31, 2022
|
|
12/2021
|
Aug. 31, 2022
|
|
01/2022
|
Sept. 30, 2022
|
|
02/2022
|
Oct. 31, 2022
|
Please note, the date printed on each vail is the manufacture date, not the expiration date. Additionally, the QR code provided on the Pfizer Pediatric Vaccine carton provides a link to the EUA but does not provide information on expiration dates.
Please continue to follow EUA recommendations for storage.
Pfizer orange cap updated fact sheet
Pfizer gray cap updated fact sheet
|
|
MODERNA COVID-19 VACCINE STANDING ORDERS FOR WASHINGTON STATE UPDATED
Recently, the Department of Health updated the Moderna COVID-19 vaccine standing orders. The standing orders now recommend booster doses of COVID-19 vaccine for all adults 18 years of age and older.
View the standing orders online.
|
|
 NEW MMWR REPORT SHOWS IMPORTANCE OF HEALTHCARE PROVIDER RECOMMENDATION FOR COVID-19 VACCINATION
In the December 17, 2021 edition of the Morbidity and Mortality Weekly Report (MMWR), the Centers for Disease Control and Prevention (CDC) reported on their telephone study which asked adults if they were recommended COVID-19 vaccine by a health care provider. The CDC found that adults who received a recommendation were more likely to have received at least one dose of COVID-19 vaccine.
You can read the full article in the MMWR online.
|
|
PREPMOD AND VACCINE LOCATOR AUTOMATION FOR PUBLIC CLINICS
A new interface was created between PrepMod and Vaccine Locator, called "Vaccinate WA." PrepMod clinics that have the "public" option selected are now populated in Vaccine Locator based on the zip code and address that PrepMod sites include with each clinic.
 |
|
Only public PrepMod clinics are shared in vaccine locator. If you have questions about Vaccine Locator or difficulty locating your clinics through the Vaccine Locator search engine, please reach out to prepmod@doh.wa.gov. |
|
|
INCLEMENT WEATHER AND RESOURCES FOR HEALTHCARE PROVIDERS
When inclement weather is expected, power outages and vaccine shipping delays are also possible.
Ways to prepare for inclement weather:
- Ensure your vaccine management plan is up to date.
- Ensure vaccines maintain stable temperatures. If you experience a power outage, please refer to the COVID-19 Temperature Excursion Guide.
- If vaccine must be moved due to an emergency, emergency transport does not require prior approval. If vaccines are transported, ensure that a digital data logger is used to record vaccine temperatures while in transit.
- Report all temperature excursions to 360-236-4223 or COVID.vaccine@doh.wa.gov, and call the manufacturers of each vaccine involved in the excursion to verify vaccine viability.
When conducting vaccination clinics prepare for a power outage by bringing paper copies of:
- Temperature logs.
- Hard copy of PrepMod registration.
- Paper to record vaccinations for data entry when system cannot be accessed.
- Vaccine Management Plan to identify if vaccines will need to be transferred to an alternate location if the facility storing the doses does not have power that will return before temperatures go out of range.
Also consider having the following items on hand incase of a power outage onsite at a clinic:
- Spare batteries
- Flashlights
Other Resources:
DOH Storage and Handling Webpage
CDC COVID-19 Vaccine Quick Reference Guide for Healthcare Professionals
|
|
|
FOR SCHOOLS AND CHILD CARES
-
COVID-19 Vaccines: Pediatric Vaccine Toolkit for Schools (PowerPoint) - This toolkit provides a variety of resources for schools, including parent handouts, backpack stuffers, and sample communication.
-
Symptom Decision Tree and Contact Tracing Checklist for K-12 Schools Updates include reference to updated quarantine recommendation, addition of contact tracing priority settings.
-
Contact Tracing Guide and FAQ for K-12 Schools Updates include updated quarantine recommendation, reference on contact tracing priority settings.
-
K-12 2021-2022 Requirements Updates include more details on contact tracing, requirements for quarantine and testing of close contacts, and requirements for performing arts performances and other school activities.
-
K-12 Schools 2021-2022 Supplemental Considerations to Mitigate COVID-19 Transmission. Updates include classroom and school closure criteria, contact tracing in high priority settings, timelines for close contact testing, and technical clarifications on antigen testing for Test to Stay programs and performing arts/sporting activities screening testing.
-
Secretary of Health’s Mask Order FAQ, to align with a technical update to the secretary’s order related to athletes and coaches at large outdoor gatherings.
School and child care guidance can be found on the Department of Health web page here.
|
|
FOR LONG TERM CARE FACILITIES
|
|
|
RESOURCES ON THE PFIZER COVID-19 VACCINE 12-15 AGE EXPANSION
In response to the authorized of Pfizer COVID-19 to children ages 12-15, the CDC has developed several useful tools for parents and clinicians to utilize.
Information for healthcare and vaccine providers provides basic information regarding the COVID-19 vaccine, consent and tools for communication to the community:
|
|
|
SIGN UP FOR THE NEWSLETTER
If you aren't already signed up for the COVID-19 Vaccine Partner Newsletter, please visit the Department of Health's email subscribers page here. Once you enter your email, on the next page expand the Immunization topic, select the COVID-19 Vaccine Partner Newsletter, and hit submit.
|
|
This newsletter summarizes content from the week of December 26, 2021 and was sent out on December 30, 2021. |
|
|
|
|