Upcoming Webinar! What Clinicians Need to Know About Available Therapeutic Options for COVID-19
On Thursday, June 16th, 2022, from 2:00PM – 3:00PM ET CDC subject matter experts will discuss, ‘What Clinicians Need to Know About Available Therapeutic Options for COVID-19’. This webinar will be focused around COVID-19 therapeutic options, including indications, efficacy, and distribution.
Webinar Overview:
- Medications that are available that can reduce chances of severe illness and death from COVID-19 infection.
- Oral antivirals that are available that can reduce chances of severe illness and death from COVID-19 infection.
- The Food & Drug Administration emergency use authorizations (EUA) for certain antiviral medications and monoclonal antibodies to treat mild to moderate COVID-19 in people who are more likely to get sick.
- The National Institutes of Health COVID-19 treatment guidelines for healthcare providers for treating COVID-19 at home or in an outpatient setting.
Interested in attending? Please visit the CDC’s Clinician Outreach and Communication Activity (COCA) webpage to access registration details and learn more.
Note: The purpose of COCA continuing education opportunities is to increase clinicians’ knowledge and competencies of threats to the public’s health, their effects on human populations, and evaluation and management strategies of these threats. COCA continuing education is a free program where providers can earn continuing education credits.
HPOP Tip! Warning for Missing Therapeutic Inventory
A warning will be displayed when selecting a product as part of placing an order when a Provider has not reported inventory for a product since it was last ordered in HPOP.
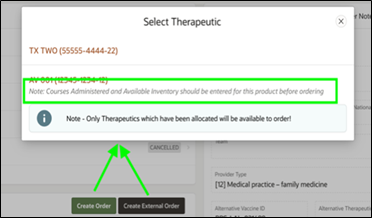 Please update inventory before placing an order to better assist in allocation determination.
For more information, please review the HPOP Provider Portal User Guide page 5-9 for reporting.
Record Therapeutics Transfer in HPOP
Providers must record any transfer of therapeutic product to another site in HPOP.
Note: This feature will not automatically ship the product; it just keeps track of the location where you plan to ship it.
1. Under Therapeutic Inventory, select the ‘Transfers’ tab.
2. Select ‘Add Transfer’ button.
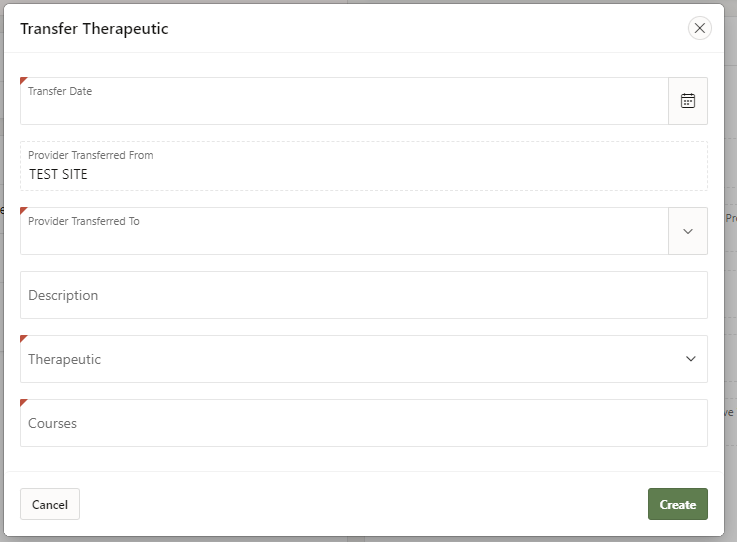
 3. Enter the required information:
- Date of transfer (select calendar icon)
- Site to which you are transferring product (search for the name of receiving facility in the drop-down)
- Therapeutic product
- Number of patient courses being transferred (record Evusheld transfers by the CARTON)
4. Select ‘Create’.
You and the receiving facility will now see the transfer under Therapeutic Inventory.
 Sites may only transfer to another location that:
- Has completed their HPOP account enrollment
- Is eligible for the type of COVID-19 therapeutic
For example, you may only transfer Paxlovid to a site that is enrolled in HPOP for antivirals.
Review the transfer therapeutic courses to a provider user guide for more information.
Note: The receiving facility must add the received therapeutics to their on-hand inventory count during their next report in HPOP.
Reminder: Guidelines for Product Return
The Office of the Assistant Secretary for Preparedness and Response (ASPR) has put together a list of guidelines to follow for Therapeutic product returns. Guidelines are as follows:
- All therapeutic products are property of the USG and must be used in accordance with EUA guidance.
- Sites of care cannot donate products to entities outside the U.S. or for use outside the U.S.
- Any returned product will be destroyed, as product integrity cannot be verified.
- Non-expired product should not be destroyed. No returns of product currently in distribution by the USG.
- All sites to first check with respective state and local health department to ensure product cannot be used/stored elsewhere in the state or region.
- Jurisdictions can also transfer product to other jurisdictions/states/territories.
- Long-term utility of authorized mAb products is expected.
- Doses discarded on site (compromised vial, unused diluted vials, etc.) should be recorded in HPOP.
- Upon these considerations, if undamaged product needs to be returned, follow the below instructions:
Reminder: CDC Health Advisory for COVID-19 Rebound After Paxlovid Treatment
On May 24th, 2022, The Centers for Disease Control and Prevention (CDC) issued a Health Alert Network (HAN) Health Advisory to update healthcare providers and the public on the potential for recurrence of COVID-19 or “COVID-19 rebound.”
COVID-19 rebound has been reported to occur between 2 and 8 days after initial recovery and is characterized by a recurrence of COVID-19 symptoms or a new positive viral test after having tested negative. The cause of a brief return of symptoms may be part of the natural history of SARS-CoV-2 infection in some persons, independent of treatment with Paxlovid and regardless of vaccination status. There is currently no evidence that additional treatment is needed with Paxlovid or other anti-SARS-CoV-2 therapies in cases where COVID-19 rebound is suspected.
The CDC reports that Paxlovid continues to be recommended for early-stage treatment of mild to moderate COVID-19.
Please review the full CDC HAN Health Advisory for more information.
|




