IMPORTANT: FDA Licenses new Rotarix liquid formulation
Michigan Dept of Health & Human Services sent this bulletin at 12/21/2022 09:54 AM EST
This message was sent to AIM, FAB, Health Systems, INE, IAP, LHD Health Officers, LHD Medical Directors, MACI, MACI 2, PH Imms Leads, MCIR Region Contacts and Imms All Staff. I apologize for any duplications
Dear Immunization Partners,
Please share the following information with providers in your jurisdiction.
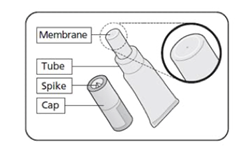
On November 4, 2022, FDA licensed a new formulation of Rotavirus Vaccine, Live, Oral (Rotarix, GSK). The new formulation is free of porcine circovirus (PCV) and comes as a liquid in a squeezable tube that does not require mixing or dilution. The single dose oral applicator increases provider convenience. Rotarix is indicated for the prevention of rotavirus gastroenteritis caused by G1 and non-G1 types (G3, G4, and G9) when administered as a 2-dose series. Rotarix is approved for use in infants 6 weeks and up to 24 weeks of age. Each dose of 1.5 mL is administered orally. See package insert for preparation and administration directions.
Both the lyophilized (requires reconstitution) and liquid rotavirus vaccine presentations will remain in use until the lyophilized formulation is used up or expires.
See the FDA Rotarix main page for more information.
CDC and MDHHS do not endorse one rotavirus product over the other.
Your Immunization Nurse Education Team,
Andrea, Dianne, Heidi, Lisa, Sarah, and Terri