COVID-19 Alaska Clinical Update
Friday, November 19, 2021
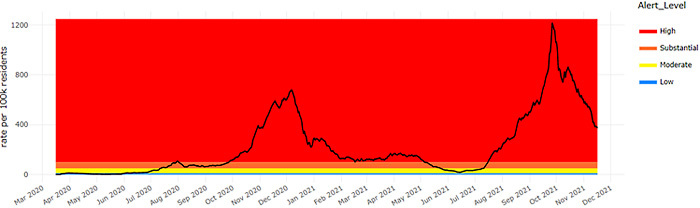
7-Day COVID-19 Case Rate – Statewide
CDC approves COVID-19 boosters for all individuals age 18 and older
On November 19, the CDC’s Advisory Committee on Immunization Practices (ACIP) voted unanimously to recommend expanding eligibility for COVID-19 boosters to all individuals age 18 and older. CDC Director Dr. Rochelle Walensky endorsed this recommendation shortly thereafter. Boosters are now recommended for all three of the COVID vaccines. Boosters for both mRNA vaccines (Pfizer-BioNTech and Moderna) are recommended 6 months after completing the initial vaccine series. Boosters for the Johnson & Johnson vaccine are recommended 2 months after receiving the initial vaccine. Individuals are still allowed to “mix and match” their primary series and booster vaccines.
CDC updates recommendations for boosters for those who received a COVID-19 vaccine outside of the US with a vaccine not authorized or approved by the FDA
On November 5, the CDC issued Emergency Use Instructions and updated its clinical considerations to allow the Pfizer-BioNTech COVID-19 vaccine to be given as an additional primary dose or booster dose to certain people who completed a vaccine primary series with certain COVID-19 vaccines that are not authorized or approved by the U.S. Food and Drug Administration. This ensures that eligible people who were vaccinated outside of the U.S. with these vaccines, or who received certain non-FDA authorized or approved COVID-19 vaccines through participation in some clinical trials, can get an additional primary dose or booster dose of Pfizer-BioNTech COVID-19 vaccine.
COVID mRNA vaccines 85% effective for preventing COVID-19 hospitalizations; unvaccinated patients accounted for 91% of deaths among COVID-19 patients in this study
In this case control study, authors evaluated the association between vaccination with mRNA COVID-19 vaccines and COVID-19 hospitalization, and, among patients hospitalized with COVID-19, the association with progression to critical disease. Authors performed a case-control analysis of 4513 adults (1983 cases with COVID-19 and 2530 controls without it) hospitalized at 21 US hospitals in 18 states between March 11 and August 15, 2021, with 28-day outcome data on death and mechanical ventilation available for patients enrolled through July 14, 2021. Among 1983 COVID-19 case patients, vaccine breakthrough patients compared with unvaccinated patients tended to be older (median age 67 vs 53 years), were more likely to be White non-Hispanic (64.0% vs 43.0%), and were more likely to be immunocompromised (40.8% vs 11.5%). Full vaccination was less common in COVID-19 case patients (15.8%) than controls without COVID-19 (54.8%) (absolute difference, −39.0%; 95% CI, −41.5% to −36.4%). Overall, COVID-19 hospitalization was strongly associated with a lower likelihood of vaccination, with an adjusted odds ratio of 0.15 (95% CI, 0.13-0.18). Among 1197 patients hospitalized with COVID-19, 142 (11.9%) were vaccinated breakthrough cases and 1055 (88.1%) were unvaccinated. Compared with unvaccinated cases, vaccine breakthrough cases were older and had more chronic medical conditions. Compared with unvaccinated cases, vaccine breakthrough cases less commonly received ICU-level care (24.6% vs 40.1%; absolute difference, −15.5%), and invasive mechanical ventilation (7.7% vs 23.0%; absolute difference, −15.3%). Unvaccinated patients accounted for 93.9% (261/278) of cases with disease progression to death or invasive mechanical ventilation. The composite of death or mechanical ventilation was experienced by 17 of 142 (12.0%) vaccine breakthrough cases and 261 of 1055 (24.7%) unvaccinated cases. Unvaccinated patients accounted for 91.0% (91/100) of deaths among patients with COVID-19 in this study. Death occurred in 9 of 142 (6.3%) vaccine breakthrough cases and 91 of 1055 (8.6%) unvaccinated patients with COVID-19.
Members of racial and ethnic minority groups had higher rates of COVID-19 infection and disease severity than White populations
In this systematic review and meta-analysis of 4.3 million patients from 68 studies, authors attempted to evaluate the association of race and ethnicity with COVID-19 outcomes and to examine the association between race, ethnicity, COVID-19 outcomes, and socioeconomic determinants. In age- and sex-adjusted analyses, African American individuals (relative risk (RR), 3.54; 95% CI, 1.38-9.07) and Hispanic individuals (RR, 4.68; 95% CI, 1.28-17.20) were the most likely to test positive for COVID-19. Following adjustment for area deprivation index (ADI), with higher values corresponding with worse socioeconomic status, African American and Hispanic individuals were almost 2 times as likely to test positive for COVID-19 as White individuals (African American: RR, 2.01; 95% CI, 1.04-3.88; Hispanic: RR, 2.09; 95% CI, 1.13-3.88), followed by Asian American individuals (RR, 1.12; 95% CI, 1.04-1.21). Asian American individuals had the highest risk of intensive care unit admission (RR, 1.93; 95% CI, 1.60-2.34). In cross-sectional studies, the combined prevalence of mortality rates were highest among African American individuals (277.15 per 1000 patients), followed by Hispanic individuals (213.34 per 1000 patients), White individuals (173.38 per 1000 patients), and Asian individuals (80.4 per 1000 patients). Decreased access to clinical care was positively correlated with COVID-19 positivity in Hispanic individuals and African American individuals. This study found a number of surprising and paradoxical correlations, such as how lower socioeconomic status was found to be associated with decreased mortality rates in Hispanic individuals in cohort studies, although the opposite result was seen in cross-sectional studies. Overall, this study provides an excellent initial review and meta-analysis of the complicated interplay between health outcomes, health disparities and social factors.
Pandemic led to reduction in life expectancy in thirty high and middle-income countries with higher than expected years of life lost in thirty one countries
In this time series analysis, researchers estimated the changes in life expectancy and years of life lost in 2020 associated with the COVID-19 pandemic in 37 upper-middle and high income countries. Reduction in life expectancy in men and women was observed in all the countries studied except New Zealand, Taiwan, and Norway, where there was a gain in life expectancy in 2020. No evidence was found of a change in life expectancy in Denmark, Iceland, and South Korea. The highest reduction in life expectancy was observed in Russia (men: −2.33; women: −2.14), the United States (men: −2.27; women: −1.61), Bulgaria (men: −1.96; women: −1.37) Lithuania (men: −1.83; women: −1.21), Chile (men: −1.64; women: −0.88), and Spain (men: −1.35; women: −1.13). Years of life lost in 2020 were higher than expected in all countries except Taiwan, New Zealand, Norway, Iceland, Denmark, and South Korea. In the remaining 31 countries, more than 222 million years of life were lost in 2020, which is 28.1 million years of life lost more than expected. The excess years of life lost were relatively low in people younger than 65 years, except in Russia, Bulgaria, Lithuania, and the US where the excess years of life lost was >2000 per 100 000.
Merck claims oral antiviral molnupiravir reduces risk of hospitalization or death by 50% compared to placebo for patients with mild or moderate COVID-19
On October 1, Merck and Ridgeback Biotherapeutics announced interim results from a Phase 3 trial investigating the safety and efficacy of their novel oral antiviral medication, molnupiravir. The planned interim analysis evaluated data from 775 patients who were initially enrolled in the Phase 3 MOVe-OUT trial on or prior to Aug. 5, 2021, and all patients had laboratory-confirmed mild-to-moderate COVID-19, with symptom onset within 5 days of study randomization. All patients were required to have at least one risk factor associated with poor disease outcome at study entry. Interim results appear to show that molnupiravir reduced the risk of hospitalization or death by approximately 50%; 7.3% of patients who received molnupiravir were either hospitalized or died through Day 29 following randomization (28/385), compared with 14.1% of placebo-treated patients (53/377); p=0.0012. Through Day 29, no deaths were reported in patients who received molnupiravir, as compared to 8 deaths in patients who received placebo. The incidence of any adverse event was comparable in the molnupiravir and placebo groups (35% and 40%, respectively). On November 4, the UK Medicines and Healthcare Products Regulatory Agency authorized molnupiravir for the treatment of mild-to-moderate COVID-19 in adults with diagnostic-confirmed SARS-CoV-2 infection and who have at least one risk factor for developing severe illness. Merck has applied to the US FDA for emergency use authorization (EUA) for molnupiravir and the FDA’s Antimicrobial Drugs Advisory Committee will meet on November 30 to discuss the application.
*These results have not gone through peer review and were announced in a press release by the sponsoring pharmaceutical companies. The results might contain errors and report information that has not yet been accepted or endorsed in any way by the scientific or medical community.
Pfizer claims novel oral antiviral reduces risk of hospitalization or death by 89% compared to placebo for patients at high risk of progressing to severe illness
On November 5, Pfizer announced interim results from a Phase 2/3 randomized, placebo controlled double-blind study of its novel oral antiviral, a combination of PF-07321332 and ritonavir, in non-hospitalized adult patients with COVID-19, who are at high risk of progressing to severe illness. The primary analysis of the interim data set evaluated data from 1219 adults who were enrolled by September 29, 2021, and enrolled individuals had a laboratory-confirmed diagnosis of SARS-CoV-2 infection within a five-day period with mild to moderate symptoms and were required to have at least one characteristic or underlying medical condition associated with an increased risk of developing severe illness from COVID-19. The scheduled interim analysis showed an 89% reduction in risk of COVID-19-related hospitalization or death from any cause compared to placebo in patients treated within three days of symptom onset (primary endpoint); 0.8% of patients who received the study drug were hospitalized through Day 28 following randomization (3/389 hospitalized with no deaths), compared to 7.0% of patients who received placebo and were hospitalized or died (27/385 hospitalized with 7 subsequent deaths). Similar reductions in COVID-19-related hospitalization or death were observed in patients treated within five days of symptom onset; 1.0% of patients who received the antiviral were hospitalized through Day 28 following randomization (6/607 hospitalized, with no deaths), compared to 6.7% of patients who received a placebo (41/612 hospitalized with 10 subsequent deaths), with high statistical significance (p<0.0001). In the overall study population through Day 28, no deaths were reported in patients who received the medication as compared to 10 (1.6%) deaths in patients who received placebo. Treatment-emergent adverse events were comparable between the study drug (19%) and placebo (21%), most of which were mild in intensity. Pfizer has applied to the FDA for emergency use authorization (EUA) but the FDA has not yet set a meeting to discuss this application.
*These results have not gone through peer review and were announced in a press release by the sponsoring pharmaceutical companies. The results might contain errors and report information that has not yet been accepted or endorsed in any way by the scientific or medical community.
Regeneron claims that single dose of its monoclonal antibody product, casirivimab and imdevimab, reduces risk of COVID-19 by 81.6% at eight months
On November 8, Regeneron announced updated results from its phase 3 trial assessing the efficacy of a single dose of its monoclonal antibody REGEN-COV (casirivimab and imdevimab) at preventing COVID-19 in uninfected individuals. The new analyses reportedly show the antibodies reduced the risk of contracting COVID-19 by 81.6% during the pre-specified 2-8 month follow-up period maintaining the 81.4% risk reduction during the first month after administration, which was previously reported. There was an 81.5% reduced risk of developing COVID-19 at any time during the 8 months after receiving REGEN-COV (20 REGEN-COV, 108 placebo; 95% CI: 70.6%,88.4%). During the 8-month assessment period, 0 individuals in the REGEN-COV group were hospitalized due to COVID-19, compared to 6 individuals in the placebo group (1 person in the first month; 5 people during months 2-8). There were no deaths due to COVID-19 in any treatment group during the 8-month assessment period. Study participants were allowed to become vaccinated if they wished once the primary efficacy treatment period (month 1) was complete. Vaccination rates during the months 2-8 assessment period were balanced, with 34.5% (n=290) of the REGEN-COV group and 35.2% (n=296) of the placebo group receiving at least 1 COVID-19 vaccine dose by the end of the 8-month assessment period.
*These results have not gone through peer review and were announced in a press release by the sponsoring pharmaceutical companies. The results might contain errors and report information that has not yet been accepted or endorsed in any way by the scientific or medical community.
Recent Calls/Presentations of Interest:
11/18 - CDC COCA Call: What Clinicians, Pharmacists, and Public Health Partners Need to Know about Antibiotic Prescribing and COVID-19 – November 18 (Link)
11/18 - AAP Town Hall: Hear the latest about COVID-19 in children. This session will begin with a brief presentation from a National Institute for Child Health and Human Development (NICHD) pediatric leader, Dr Robert Tamburro, who will provide a pediatric COVID-19 research update. The presentation will be followed by a time of facilitated Q&A.(Link to recording will be posted next week).
Previous town halls are available for viewing here
Morbidity and Mortality Weekly Reports (MMWR)
Unvaccinated persons with COVID-19 were approximately twice as likely to receive ED care or to be hospitalized than were vaccinated persons with COVID-19
In this MMWR, authors used data from Kaiser Permanente Northwest in Oregon and Washington to measure the incidence of laboratory-confirmed SARS-CoV-2 infection, emergency department (ED) visits, and hospitalizations calculated by COVID-19 vaccination status, vaccine product, age, race, and ethnicity. Among 482,464 eligible persons identified during the surveillance period, 137,616 (28.5%) were unvaccinated and 344,848 (71.5%) were fully vaccinated. A total of 7,155 laboratory-confirmed SARS-CoV-2 infections were identified, including 4,146 (57.9%) among unvaccinated and 3,009 (42.1%) among vaccinated persons. Overall incidence was 30.1 per 1,000 unvaccinated persons and 8.7 per 1,000 vaccinated persons (incidence rate ratio (IRR) = 3.5). Incidence of infection was at least three times higher among unvaccinated than among vaccinated persons; IRRs were highest among unvaccinated multiple race persons (4.3), Black persons (4.2), Asian persons (4.1), and adolescents aged 12–17 years (8.9). Among unvaccinated persons with COVID-19, 18.5% had an ED encounter, and 9.0% were hospitalized, compared with 8.1% and 3.9%, respectively, of vaccinated patients. Fifty-nine deaths occurred in unvaccinated patients, including 58 who were hospitalized; 22 deaths occurred among fully vaccinated patients, including 21 who were hospitalized. The crude mortality rate among unvaccinated persons (0.43 per 1,000) was sevenfold higher than that among fully vaccinated persons (0.06). COVID-19 hospitalizations were rare among fully vaccinated adolescents and young adults; 72% of hospitalizations among fully vaccinated persons occurred in persons aged ≥65 years. In contrast, hospitalizations among unvaccinated persons were more evenly distributed across age group with 33% among persons aged ≥65 years.
RECURRENT TOPICS
Providing COVID-19 vaccinations
All Alaskans and people who work or live in Alaska who are aged 5 years and older are eligible for vaccination against COVID-19.
If you are interested in providing the COVID-19 vaccine in your office or clinic, please visit the COVID-19 Vaccination Program Provider Enrollment page. If you have additional questions, please email Matthew Bobo at matthew.bobo@alaska.gov
Pregnancy
On September 29, the CDC released a health advisory strongly recommending COVID-19 vaccination either before or during pregnancy because the benefits of vaccination outweigh known or potential risks.
For the latest recommendations, check out the CDC webpage on COVID in Pregnant and Recently Pregnant People and recommendations from the American College of Obstetrics and Gynecology.
Monoclonal Antibodies
Monoclonal antibody treatment for COVID-19, REGEN-COV (casirivimab and imdevimab) and Eli Lilly’s bamlanivimab and etesevimab, have been approved to treat mild-moderate COVID-19 and for post-exposure prophylaxis of COVID-19 in individuals age 12 years and older who are at high risk for progression to severe COVID-19.
If you are interested in providing monoclonal antibody therapy for COVID-19 in your office or clinic, please refer to this guide from the U.S. DHSS, and then send an email to Coleman Cutchins (coleman.cutchins@alaska.gov) and CJ Kim (cj.kim@alaska.gov) for local assistance.
For the latest recommendations, check out the CDC webpage on Monoclonal Antibodies for High-Risk COVID-19 patients and COVID-19 Monoclonal Antibody Resources for Healthcare Providers.
Ivermectin
Current evidence does not support the use of ivermectin for the treatment or prevention of COVID-19.
CDC - Health advisory about ivermectin
Cochrane Review - COVID-19 and ivermectin
FDA - Why you should not use ivermectin to treat or prevent COVID-19
Infectious Disease Society of America (IDSA) guidelines do not recommend ivermectin
Post-acute Sequelae of COVID-19 (PASC)
For the latest recommendations, check out the CDC webpage on Post-COVID-19 Syndrome and Evaluating and Caring for Patients with Post-COVID conditions
Myocarditis
For the latest recommendations, check out the CDC webpage on myocarditis and COVID-19 vaccines
COVID-19 Speakers’ Bureau
Anyone can request a free presentation for a group interested in learning more about the COVID-19 vaccines available in Alaska.
Aside from COVID-19
CDC updated Blood Lead Reference Value as result of decreasing lead levels among children, however more children will now be identified as having elevated blood lead levels and will receive recommendations for follow-up services
On
Upcoming Events/Conferences/Presentations
Drug Addiction Treatment Act (DATA) Waivers ECHO beginning November 10
UAA Center for Human Development and the State of Alaska Division of Behavioral Health are pleased to offer a new Project ECHO for medical providers who are currently DATA waivered and/or medical providers who are interested in obtaining a DATA waiver.
|
Date
|
Time
|
Session
|
| Dec 8 |
12:00 p.m. |
Buprenorphine pharmacology |
| Jan 12 |
12:00 p.m. |
Preparing for your first OUD MAT visit |
| Feb 9 |
12:00 p.m. |
Successful strategies for buprenorphine home inductions |
| Feb 23 |
12:00 p.m. |
Using extended-release monthly buprenorphine |
| Mar 9 |
12:00 p.m. |
Microdosing/macrodosing buprenorphine |
| Apr 13 |
12:00 p.m. |
All things UDS/oral fluid tests for monitoring |
| May 11 |
12:00 p.m. |
Behavioral Health - Psychosocial adjuncts to MAT |
| Jun 8 |
12:00 p.m. |
Polysubstance use |
| Jul 13 |
12:00 p.m. |
Tapering or stopping buprenorphine treatment |
| Aug 10 |
12:00 p.m. |
Prenatal/OB MAT |
| Sep 14 |
12:00 p.m. |
Understanding and reducing buprenorphine diversion |
Registration link for DATA Waivers ECHO
Hosted and facilitated by UAA’S Center for Human Development Alaska ECHO Project in partnership with the State of Alaska Division of Behavioral Health. For questions, please contact echo@alaskachd.org
ANTHC Tribal Health Webinar Series
The ANTHC Tribal Health Webinar series occurs on Friday from 12-1pm on Zoom and is open to the public. Here is the upcoming schedule for the fall and the Zoom link.
|
Date
|
Session
|
| 11/19 |
Katie Presser, PharmD: Statewide Antimicrobial Stewardship |
| 12/03 |
Marah Gotcsik, MD: ANMC Guideline on Preterm Infant Nutrition through 2 Years Old |
| 12/10 |
Leslie Kerzner, MD: Care of the NICU Grad for Primary Care Providers |
| 01/14 |
Hope Finkelstein, Alaska FASD Program Manager: DHSS FASD Program |
| 01/21 |
Victoria Miller, MFA, LPC: Trauma Informed Care Mini-Series. Part 1: Early Life Adversity, Later Life Disease & Implications for Health Care |
| 01/28 |
Amber Frasure, MS: Trauma Informed Care Mini-Series. Part 2: A Deeper Dive into the Signs and Symptoms of Psychological Trauma and Specific Strategies to "Trauma Inform" your Medical Practice |
| 02/04 |
Amber Frassure, MS: Trauma Informed Care Mini-Series. Part 3: Effectively Communicating with Patients Using Motivational Interviewing |
| 02/11 |
Mary Schneider, BS, REHS: State of Alaska Lead Surveillance Program |
| 02/18 |
Vasisht Srinivasan, MD: Updates in Stroke Management |
Join Zoom Meeting
https://anthc.zoom.us/j/98667611681
Meeting ID: 986 6761 1681
One tap mobile
+16699006833,,98667611681#
Use of Valid Background Check Clearances by Multiple Employers
Providers who have applicants with pending background checks with DHSS BCU may allow individuals to start work prior to the Background Check Program completing their check IF the applicant has documented proof of having a current Alaska DHSS background check (provisional or complete) for another DHSS provider. The applicant must provide their new employer a copy of the final background check eligibility letter for the other provider, and providers are required to maintain a copy of that eligibly document in the employee’s file. Providers who hire employees in this manner MUST provide supervised access to those employees, i.e., to ensure the protection of recipients of services, the provider must maintain a prudent level of awareness of the whereabouts of an individual for whom supervised access is required. Employees working in this way may NOT be the only individual working in a facility or a setting, as another employee must be present to provide the supervised access. The provider’s oversight agency will monitor providers to ensure they are in compliance with these requirements, and providers must submit proof of compliance to those agencies upon request. Questions? Please contact your DHSS oversight division.
CDC Clinical Support: There is a Clinician On-Call Center, a 24-hour hotline with trained CDC clinicians standing by to answer COVID-19 questions. Call 1-800-CDC-INFO (800-232-4636) and ask for the Clinician On-Call Center.
The most up-to-date, evidence based COVID-19 treatment guidelines can be found at:
NIH COVID-19 Treatment Guidelines
IDSA Guidelines on the Treatment and Management of Patient with COVID-19
Alaska Responders Relief Line (844) 985-8275
Your well-being matters. Your behavioral health colleagues are standing by to talk 24/7: 844-985-8275
Recognizing the unique stressors that providers face, the Division of Behavioral Health has established a 24/7 support line, (844) 985-8275, for healthcare and behavioral professionals impacted by COVID personally and professionally. Staff supporting the call line recognize callers are often first responders and will allow callers to openly express their experiences and feelings serving Alaskans impacted by COVID.
This service is also available to immediate family members of first responders who may be experiencing stress, anxiety and other hard to label emotions as a result of their loved one engaging on the front lines.
AK Clinical Reminders — UPDATED November 19, 2021

These ECHO sessions are produced and facilitated by
UAA’s Center for Human Development Alaska ECHO project
in partnership with the State of Alaska, Department of Health & Social Services
Session information and recordings of previous ECHO sessions
subscribe to ECHO calendar updates | email: echo@alaskachd.org | website: akecho.org
Important Note: Most Division of Public Health ECHO series will be taking a break the week of November 22 for Thanksgiving and from December 20-December 31 for the winter holidays. Please check ECHO registration pages for additional information on holiday scheduling.
This month’s Local Government ECHO is cancelled for the Alaska Municipal League’s annual local government conference. In lieu of an ECHO, AML is hosting a conversation with the State’s public health team at the Alaska Conference of Mayors meeting on the 18th. We look forward to seeing attendees there. The next Local Government ECHO will be December 15th.
Our Alaska Medical Providers ECHO is returning to a once-monthly schedule, on the 1st Tuesday of the month.
Upcoming Session Highlights:
Alaska Perinatal ECHO
Monthly on 3rd Thursday from 6-7 p.m. Register
Next Session: November 18th
Please join us for the next Perinatal ECHO on Preventing Preterm Births in Alaska on November 18th 6:00-7:00pm! This session will feature a didactic presentation by Dr. Jodi Abbott, Maternal Fetal Medicine Physician and Director of Obstetrics and Gynecology at Boston Medical Center and Associate Professor of Obstetrics and Gynecology at Boston University School of Medicine. This session will also include Alaska preterm birth data presented by Margaret Young, State of Alaska MCH Epidemiology Unit Manager and a case presentation by Dr. Ellie Hogenson, OB/GYN at Fairbanks Memorial Hospital. As always, we will have ample time for Q&A and discussion with presenters and the hub team.
Regular Series
Alaska Medical Provider ECHO (formerly COVID-19 for AK Healthcare Providers)
Monthly on the 1st Tuesday of the month from 7-8 pm
This ECHO aims to increase knowledge and share best practices among medical providers across Alaska as well as to increase access to information for those living in rural areas of the state.
School Health ECHO
Every Monday from 3:00-4:00 p.m Register
The School Health ECHO is a virtual learning network intended for professionals in the education setting (administrators, school-based nurses, etc.) to interface with a team of medical and education experts in Alaska.
Vaccine ECHO for Providers
Weekly on Tuesday from 2-3 p.m. Register
The Vaccine ECHO for providers provides planning and operation updates to vaccine providers across Alaska, while answering any questions you may have.
Palliative Care ECHO
Monthly on the first Tuesday from 12-1 p.m.
This monthly ECHO is for all Alaskan healthcare disciplines supporting patients with serious illness, and aims to provide support and up-to-date information regarding Palliative Care during this period of COVID-19. Our next session will include a didactic presentation Meeting People Where They Are: Providing Palliative Care to Rural Populations by guest presenter Nancy Joiner APRN, ACHPN from the University of North Dakota’s Center for Rural Health.
Public Science ECHO
Weekly on Wednesday from 12-1 p.m. register via Zoom
The Alaska Public Health Science ECHO is a virtual learning network intended for the general public to interface with our Public Health Leadership Team to explore the science of the COVID-19 virus, other public health topics, and current best practices. Or view via concurrent livestream to Facebook: https://www.facebook.com/akechoprograms
Local Government Public Health ECHO
Monthly on the third Wednesday from 3-4 p.m. Register
The Local Government Public Health Analysis ECHO is a virtual learning environment intended for local Alaska government leaders to interact with the State Public Health Leadership team and focuses on pandemic mitigation tools available, and how to use them.
Long Term Care Facilities ECHO
Second Wednesday of the month from 4-5 p.m. Register
Please join other staff and administrators of Alaska’s assisted living homes and residential care facilities to gather, learn, share, and grow. COVID has disrupted the fabric of our daily life and we can always learn from each other as we adjust to the new normal.
Healthcare Specific Situational Awareness ECHO
Weekly on Thursday from 12-1 p.m. Register
The Healthcare Specific Situational ECHO is a virtual learning network intended for healthcare professionals to interface with our Public Health Leadership Team to explore current best practices and the most recent information related to Public Health.
Perinatal ECHO
Monthly on 3rd Thursday from 6-7 p.m. Register
The Alaska Perinatal ECHO is a virtual learning network intended for medical providers caring for pregnant patients and their newborns.
EMS ECHO
Monthly on the 1st and 3rd Wednesday 1-2 p.m Register
The EMS ECHO is a virtual learning network intended for Emergency Medical Services and related personnel in Alaska to amplify best practices. Sessions are topic-driven and typically include a guest presenter or a brief lecture with an interactive case or process discussion.
Co-Occurring Behavioral Health, Opioid and Stimulant Use Disorders ECHO Returning with new didactics!
1st & 3rd Wednesdays from 12:00pm-1:00pm, November 3, 2021 - April 20, 2022
The Co-Occurring Behavioral Health (COBH), Opioid and Stimulant Use Disorders ECHO facilitates a virtual network for behavioral health providers to learn best practice care through real-time access to experienced subject matter experts and their peers. Each session includes a brief lecture, de-identified case presentation for participants to receive feedback on complex cases, and open discussion facilitated by an interdisciplinary team.
Data Waivers ECHO New ECHO!
2nd Wednesdays from 12:00pm-1:00pm, November 10, 2021 - September 14, 2022
The Data Waivers ECHO facilitates a virtual network for clinical providers to learn and implement best practices for offering buprenorphine and other medically assisted treatment (MAT) services under a data waiver. Each session includes a brief lecture, de-identified case presentation for participants to receive feedback on complex cases, and open discussion facilitated by an interdisciplinary team.
|
COVID-19 testing guidelines and test site locator
AK COVID-19 clinical hotline for physicians: 833-751-4212. Staffed 24/7.
8PM-8AM is for urgent/emergent questions only.
AK Responders Relief Line: 24/7 behavioral health for everyone working in healthcare
during the COVID-19 pandemic: 1-844-985-8275
|



