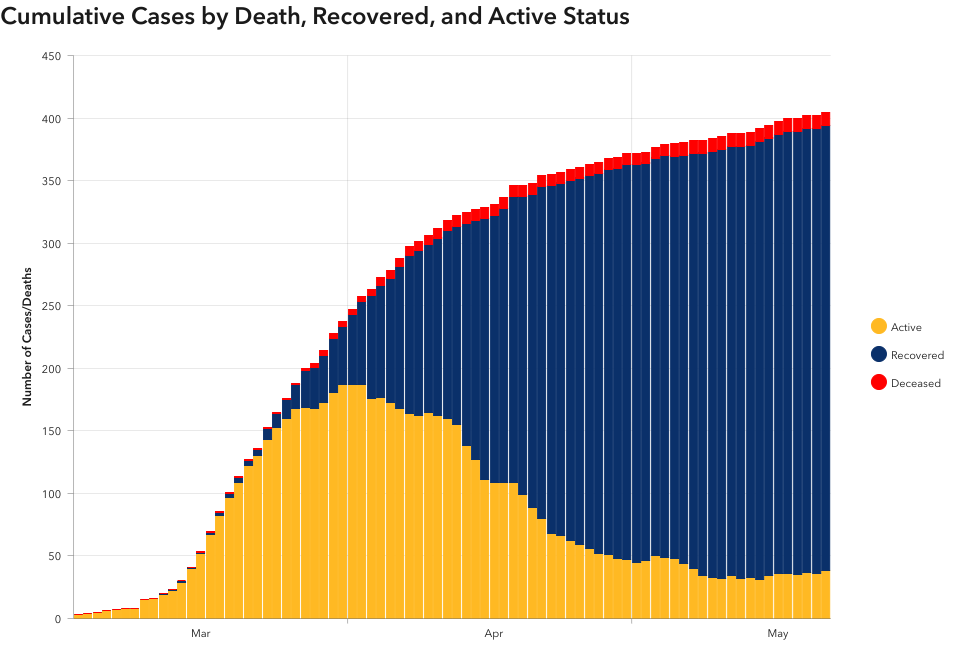
COVID-19 Alaska Daily Clinical Update, May 25
Alaska DOH sent this bulletin at 05/25/2020 08:36 PM AKDT
Monday, May 25, 2020
Liz Conway Ohlsen, MD
With information from Anne Zink, MD, FACEP, Alaska Chief Medical Officer
|
A study of 16 placentas from patients who had COVID-19 during pregnancy found an unusually high occurrence of perfusion problems and clots compared to placentas examined before the pandemic. In particular, a high prevalence of maternal vascular malperfusion, decidual arteriopathy and intervillous thrombus compared to controls was found. Only ten of the 16 women had symptoms at any time associated with COVID-19 and none became severely ill.
15 women had live births in the third trimester (14 at term, one at 34 weeks), and one delivered in the second trimester after intrauterine fetal demise; the last of which was excluded from broader statistical analysis.
Only one patient was hypertensive. This pattern of placental injury is also seen in hypertensive disorders of pregnancy, and authors suggest that these changes in the placenta may reflect a systemic inflammatory or hypercoagulable state. While this is insufficient evidence to justify major changes in clinical practice, increased antenatal surveillance may be warranted for pregnant women with COVID-19.
A preliminary report from a double-blind, randomized, placebo-controlled trial of IV remdesivir in adults hospitalized with COVID-19 pneumonia was unblinded early on the basis of an analysis showing shortened time to recovery in the remdesivir group. Of 1059 patients, the 538 assigned to remdesivir had a median recovery time of 11 days (95% CI 9-12 days) while the 521 assigned to placebo had a median recovery time of 15 days (95% CI 13-19 days). Mortality at 14 days was also lower in the group randomized to remdesivir (7.1% vs 11.9%) but these results were not statistically significant- hazard ratio for death 0.7 (95% CI 0.47-1.04). Benefit seemed to be least pronounced among patients requiring mechanical ventilation or ECMO at time of enrollment, with nearly the same rates of recovery whether a patient received remdesivir or not. While remdesivir is still currently only recommended as part of a clinical trial, we may see use expand in the coming weeks on the basis of these data. These data suggest that while remdesivir may not help once patients are critically ill, it may shorten the hospital stay of patients who receive the drug. More data may help distinguish whether there is also a mortality benefit in patients when started before the patient develops critical illness.
|