Fluorescent Polarization Assay (FPA) now being offered at the ADDL
 ADDL is now offering a rapid test method for the screening and confirmation of samples for Brucella abortus/suis in bovine and porcine serum. The FPA is a qualitative antibody test that uses fluorescent polarization technology to rapidly detect the presence of antibodies indicative of prior infection with Brucella abortus/suis, or vaccination with a smooth strain vaccine such as Strain 19.
Fluorescent polarization has been in use for years in human medicine, and now has been adapted for use in veterinary diagnostic laboratories and in the field for the detection of brucellosis in cattle and swine. The test is based on the premise that larger molecules will spin at a slower rate than smaller molecules in a solution. When a fluorescent dye-tagged antigen is introduced to a sample serum in solution, it will search out and combine with the target antibody to form an antigen-antibody complex that results in a larger molecule that spins at a slower rate. A beam of polarized light can then determine the rate of spin on the molecules in the solution and will compare it to the rate of the negative control, generating a numbered result incremented in millipolarization (mP) units. The greater the difference between the negative and the sample, the higher the number generated. Numbers greater than 20 mP are considered to be positive, whereas those falling below 10 mP are considered negative. Numbers falling between 10 – 20 mP are regarded as suspect.
The advantages of the FPA are many, making it a highly desirable testing platform to implement in the laboratory. It is simple to perform, gives rapid results, and is extremely reproducible across laboratories and instruments. And more notably, it reduces human error and the variability that occurs when reading existing test methods. The entire assay is done in solution, in a single tube with no precipitation or washing steps and can be read in minutes. It is also USDA and OIE approved and does not react with RB 51 vaccinates. Our analysts have been proficiency tested on NVSL protocol to ensure quality results and are ready to serve your needs in the ADDL.
|
 Regulatory Brucella canis Testing
A new program implemented by the Ohio Department of Agriculture outlines testing standards and procedures to become certified as a B. canis-free high volume dog breeder. ADDL B. canis sample submission instructions and test fees can be viewed here. High volume breeders must test their canines regularly to gain certification as a B. canis-free kennel. The new regulations in Ohio will reduce and hopefully eliminate outbreaks of B. canis in our state.
Brucella canis is a dangerously contagious and infectious microorganism. This bacterium is responsible for abortion, reproductive impairment, and infertility in dogs, as well as ocular inflammation. Additionally, the disease is zoonotic which means it can be transmitted to humans. Humans infected with B. canis may experience the following symptoms: fever, fatigue, headache, weakness, malaise, chills, sweats, weight loss, hepatomegaly, splenomegaly, and lymphadenopathy.
|
 New at ADDL: Next Generation Sequencing
ADDL acquired a state-of-the-art high-throughput Illumina MiSeq instrument in 2014 to perform next generation sequencing (NGS) of samples in support of surveillance programs, subtyping, and discovery of novel or emerging livestock pathogens. Using NGS, we have identified variant PRRS virus and novel porcine coronaviruses circulating in the field. This technology will help us identify new and emerging pathogens, such as the variant PED virus that we identified in 2013. We offer whole genome sequencing of bacteria and viruses.
|
Case Report: Goat kid lesions lead to FMD, VS viruses investigation
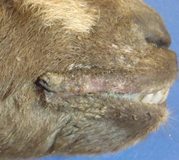 History: A 9-week-old Pygmy doe kid was presented for necropsy at the ADDL as a possible foreign animal disease investigation to rule out infections with foot and mouth disease (FMD) and vesicular stomatitis (VS) viruses. The owner brought the kid and its sibling from out of state without a certificate of veterinary inspection (CVI). Both kids developed erosive lesions on the oral cavity and muzzle, some covered in fly eggs. The animal was thin and dehydrated, and it died en route to a veterinary clinic 11 days later. A littermate had a temperature of 104.7 F and also had crusty lesions around the muzzle.
|
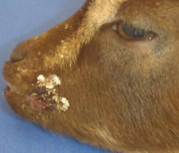 Investigation: Necropsy showed multiple nodular, raised, firm 3-5 mm lesions and erosions at the commissures of the lips. Samples of the lip lesions were submitted to the ADDL for histopathology and electron microscopy, and were submitted to the Plum Island Foreign Animal Disease Diagnostic Laboratory (FADDL) for virus isolation and PCR testing to rule out viruses mentioned above.
|
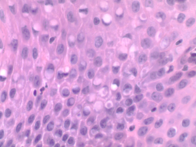 Diagnosis: A histologic diagnosis of lymphoplasmacytic and proliferative dermatitis with eosinophilic intracytoplasmic inclusions was made at the ADDL. Note viral inclusions within a section of hyperplastic epidermis.
Virus isolation at Plum Island was positive, and PCR testing of the supernatant was positive for parapox virus nucleic acid, and was negative for nucleic acid of FMD and VS viruses. The final diagnosis was parapox infection (contagious pustular dermatitis, infectious labial dermatitis, soremouth, scabby mouth, contagious ecthyma).
|
Lesson: Bringing
animals into Ohio without a certificate of veterinary inspection (CVI) is a
violation of state law (OAC 901:1-17-01 (A)&(C) as well as
901:1-17-06(A)(1)&(2). The private practitioner did well to notify the Ohio
Area District Director office (614-856-4735) of this animal movement violation,
and to help initiate a foreign animal disease investigation when two of the
imported animals had fever and oral lesions. This resulted in a rapid diagnosis
of a domestic disease (contagious ecthyma) between the Ohio ADDL and USDA
FADDL, which ruled out the presence of a foreign animal disease. Parapox virus infections in sheep, goats and
cattle can cause painful lesions on the hands and arms of humans. Sanitation
and hygiene measures during and after handling affected animals are important
to avoid human illness.
Laboratory Updates
Molecular Diagnostics
We now offer same-day test results using molecular assays that detect nucleic acid from Mycoplasma hyopneumoniae, porcine enteric coronaviruses (including PEDV), PRRS virus, swine influenza virus and avian influenza virus. To see our comprehensive list of rapid PCR tests and recommended sample types, check out the ADDL Tests and Fees page.
Our molecular diagnostics staff are certified by the National Animal Health Laboratory Network to perform rapid real-time PCR testing for detection of classical swine fever virus, foot-and-mouth disease virus, Newcastle disease virus, and avian influenza virus. In the event of an outbreak, our array of real-time PCR machines allows for high-throughput testing of >1000 samples in a regular 8 hour work day.
Pathology
The Pathology Section hosted two, 3-hour wet labs for the national Turkey & Broiler Health Layer Health Management School conducted this year at the Ohio Department of Agriculture campus on May 4 and 5, 2015. In the last five years, wet labs have also been conducted on the necropsy floor by ADDL pathologists for other poultry veterinarians, for sheep producers, for goat producers, and for fish producers and veterinarians interested in aquaculture.
Caprine Arthritis Encephalitis Virus AGID no longer offered
The CAEV AGID test will no longer be offered at the Ohio ADDL. The sole manufacturer of this test kit has chosen not to license new test kit lots with the USDA. Since the ADDL only uses USDA-approved, licensed test kits, the CAEV AGID has been removed from the Serology Section schedule of tests.
The alternative to using the AGID test is to use the currently offered Small Ruminant ELISA test – which includes the detection of CAEV and Ovine Progressive Pneumonia Virus (OPPV). After many years of comparison between the ELISA and the AGID for CAEV, the laboratory has observed a very high percentage of agreement between the two tests. Based on this in-house comparison, we feel confident in using the ELISA as an effective replacement for the AGID at this time. Should a new CAEV AGID become available and licensed, the laboratory will announce the re-addition to the schedule of tests.
|